Capone statutes of Experimental C19 Gene Vaccine's;
42 U.S. Code § 262 - Regulation of Biological Products..
This states under (3)(b).
"No person shall mislabel or misbrand a package or container of a biological product or alter a label or marking on the package or container of the biological product so as to falsify the label or marking.
Combined with;
21 U.S. Code § 331 - Prohibited acts - The following acts and the causing thereof are prohibited:
(a)The introduction or delivery for introduction into interstate commerce of any food, drug, device, tobacco product, or cosmetic that is adulterated or misbranded.
The definition of a common vaccine is:
Vaccine is a preparation used to stimulate the immune system so that the person being vaccinated becomes immune without undergoing disease.
The definition of a Genetic Vaccine is:
Gene vaccines are a new approach to immunization and immunotherapy in which, instead of a living or inactivated organism (or a subunit thereof), one or more genes encoding the pathogen's proteins are delivered.
The goal of this approach is to generate immunity to diseases for which traditional vaccines and treatments have not worked, to improve vaccines and to treat chronic diseases.
Gene vaccines use advances in immunology and molecular biology to more specifically tailor immune responses (cellular or humoral, or both) to selected antigens. They are still under development in research and clinical studies.
The difference is that a regular vaccination is well-proven, and does not change any of your body's genes or cell structure.
And is known and discussed under the name: Vaccine.
But a Genetic Vaccine is a form of Gene Therapy.
Which is defined as:
Gene therapy is the transfer of genetic material to cells to prevent or treat disease, or to affect biological functions. Different types of gene therapy are under development, and these can provide new treatment options for a number of different diseases and disorders.
Therefore, these are also referred to as Genetic Vaccines by the industry.
See:
https://genetic-vaccine-development.com/
Documents recived from Sciex, when i enrolled in their program to help advance my Genetic Vaccine's.
(I don’t have such a program but they don't know that)
Furthermore:
The very definition of the Category Genetic Vaccine clearly states that there is no known approved Genetic Vaccine.
So you have a new Experimental Category and New Experimental products in that category.
It does not get more experimental than that!
To easy explain the difference to anyone:
(To explain it another way: You can call a wrench, a socket wrench as much as you want. But it never becomes a socket wrench. Although it can do the same job, they are still two completely different tools.)
(An even better comparison is a fossil-fueled car vs an electric car. Both can look almost identical on the outside, but everything else is different in the build-up. It's the exact same difference between a regular Vaccine and a Genetic Vaccine, they look the same on the outside but they are not the same in any way)
This proves;
The Authorization is wrongly written for a Vaccine & Not a Genetic Vaccine.
The Product description in the purchase agreement is wrong / product received & given.
This in term is;
Wilfull Misconduct - Kills the authorization and purchase agreement contracts.
Material Breach of Contract;
The Product received is not a vaccine, it's a genetic vaccine & gene therapy.
All this is Bio Terrorisme which violates UN Security Resolution 1373.
Bioterrorism is terrorism involving the intentional release or dissemination of biological agents. These agents are bacteria, viruses, insects, fungi or toxins, and may be in a naturally occurring or a human-modified form, in much the same way as in biological warfare.
Links;
42 U.S. Code § 262 - https://t.co/RKZD8ioTju
21 U.S. Code § 331 - https://t.co/TytgK867wX
Willfull Misconduct;
Material Breach Of Contract;
Bio-Terrorism defined;
UN 1373;
Eu;
EudraLex:
Document;
CELEX_32011L0062_EN_TXT.pdf
Link: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32011L0062
33. Falsified medicinal product:
Any medicinal product with a false representation
of:
(a) its identity, including its packaging and labelling, its name or its composition as regards any of the ingredients including
excipients and the strength of those ingredients;
Norwegian principles on labeling medical products;
1;
GENERAL PRINCIPLES
Scope
Labelling of medicinal products (packaging) is essential for safe and proper use of the medicinal product by the patients and healthcare professionals.
2;
The approval of the labelling information is part of the authorisation process for all medicinal products.
- Note we follow the same eu law and directive.
So this should also be relevant in the EU.
____________________________________________________________________________
Viral Vector & RNA Vaccine's is under the gene therapy category - Vector Therapy.
____________________________________________________________________________
EMA Guidelines for vector therapy:
__________________________________
FDA Guidelines:
____________________________________________________________________________
American Society Of Gene & Cell Therapy had this to say;
____________________________________________________________________________
Informed Consent:
HHS regulations at 45 CFR 46.116 state that no investigator may involve a human being as a subject unless the investigator has obtained the legally effective informed consent of the subject or the subject's legally authorized representative.
https://www.hhs.gov/ohrp/regulations-and-policy/guidance/faq/informed-consent/index.html
Valid informed consent for research must include three major elements:
(1) disclosure of information,
(2) competency of the patient (or surrogate) to make a decision, and
(3) voluntary nature of the decision. US federal regulations require a full, detailed explanation of the study and its potential risks.
14. jun. 2021
___________________________________
Gene Vaccine's in use:
Messenger (RNA Vaccine):
Moderna’s mRNA-1273 vaccine study:
https://clinicaltrials.gov/ct2/show/NCT04470427
Actual Study Start Date :
July 27, 2020
Estimated Primary Completion Date :
October 27, 2022
Experimental: mRNA-1273
Biological: mRNA-1273
Sterile liquid for injection
Pfizer & BioNTech’s mRNA Vaccine Study:
Actual Study Start Date :
April 29, 2020
Estimated Primary Completion Date :
May 2, 2023
Experimental: 10 µg dose, 18-55 years of age (2 doses)
Biological: BNT162b1
Intramuscular injection
https://clinicaltrials.gov/ct2/show/NCT04368728
____________________________________________________________________________
Viral Vector Vaccine:
AstraZeneca Study of AZD1222
Actual Study Start Date :
August 28, 2020
Estimated Study Completion Date :
February 14, 2023
Experimental: AZD1222
Approximately 20,000 participants randomized to the AZD1222 arm
Biological: AZD1222
AZD12222 is a recombinant replication-defective chimpanzee adenovirus expressing the SARS-CoV-2-5 surface glycoprotein.
https://clinicaltrials.gov/ct2/show/NCT04516746
____________________________________________________________________________
Janssen’s Ad26.COV2.S Investigational Vaccine Study:
Actual Study Start Date :
September 7, 2020
Estimated Study Completion Date :
January 2, 2023
Experimental: Ad26.COV2.S
Biological: Ad26.COV2.S
Ad26.COV2.S will be administered at a single dose of 5*10^10 virus particles (vp) on Day 1 (or Month 6 for placebo recipients).
Other Names:
JNJ-78436735
Ad26COVS1
https://clinicaltrials.gov/ct2/show/NCT04505722
____________________________________________________________________________
Novavax NVX-CoV2373 Investigational Vaccine Clinical Study:
Actual Study Start Date :
December 27, 2020
Estimated Primary Completion Date :
June 30, 2023
Experimental: SARS-CoV-2 rS/Matrix-M1 Adjuvant
Biological: SARS-CoV-2 rS/Matrix-M1 Adjuvant (Initial Vaccination Period)
Alternating intramuscular (deltoid) injections of SARS-CoV-2 rS co-formulated with Matrix-M1 adjuvant (0.5 mL) on Days 0 and 21 in Initial Vaccination Period.
Other Name: NVX-CoV2373
https://clinicaltrials.gov/ct2/show/NCT04611802
____________________________________________________________________________
Links of proof:
https://genetic-vaccine-development.com/
____________________________________________________________________________
https://genetic-vaccine-analytical-development.com/
____________________________________________________________________________
https://www.humanvaccinesproject.org/
____________________________________________________________________________
Book on Gene Theraphy Vol 1:
____________________________________________________________________________
Don't want to use the link:
Type in oligonucleotide compendium in Google search and download the Sciex pdf file.
____________________________________________________________________________
Pages from the book of gene therapy;
____________________________________________________________________________
More on vector therapy;
Link to Vector Therapy summit:
https://next-gen-genetherapy-vectors.com/
____________________________________________________________________________
(Because they refused to look at the bodies and damage by this unholy abomination's)
(No body no crime)
However these statutes are broken therfore criminal intent from the start is provable.
Every Authorization is broken, and all purchase agreements are voided.
Due to material breach of contract.
All of this becomes Bio Terrorisme in itself, but combined with the death and destruction, not to mention the most unholy act upon god's living tempels. (all humans are a living tempel of God)
Easy the worst evil act in human history.
____________________________________________________________________________
Summit’s notes and people of interest;
From 2021 May - Genetic Vaccine Summit;
____________________________________________________________________________
Title: Genetic Vaccine Development for Infectious Diseases Summit
Slogan:
Be at the Forefront of the New Era
of Vaccinology to Prevent,
Treat, and Cure Infectious Diseases
Worldwide.
Speakers:
Lidia Oostvogels
VP Area Head
Infectious
Diseases
CureVac
Johan Vekemans
Global Clinical
Head SARS-CoV-2
Vaccine
AstraZeneca
Jacqueline Miller
Sr. VP Infectious
Disease
Development
Moderna
Philip Dormitzer
Vice President &
Chief Scientific
Officer: Viral
Vaccines
Pfizer
Kate Broderick
Sr. VP Research &
Development
INOVIO
Andreas Kuhn
Sr. VP RNA
Biochemistry &
Manufacturing,
BioNTech
Intro:
With pandemic
vaccines now launching at record
speeds, the first Genetic Vaccine
Development for Infectious Diseases
Summit will focus on overcoming the
translational and clinical challenges of
developing genetic vaccines targeting
infectious diseases, including Influenza,
Lassa fever, Ebola, MERS, HIV, Zika, and
COVID-19.
With a focus on mRNA, DNA, and
engineered viral vector-based vaccines,
this niche 3-day agenda has been
specifically designed to help industry
experts to:
Progress your own expertise
and experience while forging
complementary partnerships and
alliances Overcome immunogenicity challenges
and design safe and efficacious vaccine
formulations Define clinically meaningful endpoints
to successfully develop and safely
deliver optimal doses for public use.
___________________________________________________________________
Accelerating next-generation vaccine
development for unmet and urgent
needs for all infectious diseases
Welcome to the Inaugural
Genetic Vaccine Development
for Infectious Diseases Summit
Understand how the optimization of
lipid nanoparticles allows for efficient
and safe delivery of mRNA
Hear Moderna speak on the use of
their mRNA technology platform for the
development of mRNA-based vaccines,
and a case study on mRNA-1273, one of
the first approved COVID-19 vaccines
___________________________________________________________________
Learn the critical steps in mRNA
vaccine development that allowed for
the ‘light speed’ deployment of a SARSCoV-
2 vaccine
From pre-clinical studies to large-scale
manufacture, BioNTech will be sharing
how they went about developing the first
COVID-19 vaccine, as well as sharing preclinical
data on wider infectious disease
programs
___________________________________________________________________
Optimizing next-generation vaccine
development and lessons learned from
COVID-19
Take part in an interactive panel
discussion with Pfizer and AstraZeneca
to learn how new vaccine technologies
can stave off future outbreaks, and how
the future of vaccine development has
been changed by COVID-19.
___________________________________________________________________
Discover the latest updates in DNAbased
vaccines against infectious
diseases
Hear INOVIO speak on efficacy in
animal challenge models, thermostability
of DNA vaccines, and clinical
immunogenicity and safety profiling
of INO-4800 in the race to be the first
approved DNA vaccine for human use
___________________________________________________________________
CEPI:
Clinical and Operational Challenges of
COVID-19 Vaccine Development and future
opportunities
Understand how defining clinical trial
endpoints for a new pathogen with limited
pre-existing evidence can be overcome, why
we need correlates of protection, and how
to establish vaccine efficacy when placebocontrolled
trials become more difficult.
___________________________________________________________________
Speakers and topics:
Kathleen Neuzil
Professor of Medicine
and Paediatrics
University of
Maryland School of
Medicine
9.00 Update on Clinical Trials Involving Pregnant Women with
COVID-19 Vaccines
___________________________________________________________________
Stanley Plotkin
Co-founder of
CEPI and Professor
Emeritus of Virology
University of
Pennsylvania
9.30 Controlled Human Infection Models to Further Validate the
Efficacy of COVID-19 Vaccines
• Advantages of CHIM trials to determine whether prior infection is
protective and to establish what immune responses lead to that
protection
• Identification of correlates of protection to be used to infer the likely
efficacy or lack of efficacy of vaccine candidates
___________________________________________________________________
Andreas Kuhn
Senior VP, RNA
Biochemistry &
Manufacturing
BioNTech
10.00 Development of an mRNA-based COVID-19 Vaccine at
‘lightspeed’: From Pre-Clinical Studies to Large-scale
Manufacture
- Side note; "Lightspeed" is a reference to Operation Warpspeed"
• Understanding how mRNA is a novel therapeutic platform technology
• Developing insight into how mRNA is suited for individualized
approaches and for rapid development of vaccines against newly
emerging infectious diseases
• This could be demonstrated with the development of a vaccine against
SARS-CoV-2 in less than a year
___________________________________________________________________
Jacqueline Miller
Sr. VP Infectious
Disease Development
Moderna
10.30 Moderna’s COVID-19 Vaccine: A Summary of Available Clinical
Data
• Through the collaboration with government and academia, the
Moderna COVID-19 vaccine went from initiation of clinical development
in March, 2020 to initial authorizations in December 2020 while
conducting Phase 1, Phase 2, and Phase 3 studies safely and in parallel
____________________________________________________________________________
Side note; Slides - From the Document:
Vaccines and Related Biological Products Advisory Committee October 22, 2020 Meeting Presentation- COVID19 Vaccine
Development- NIH Role.pdf
____________________________________________________________________________
____________________________________________________________________________
• Vaccine efficacy against symptomatic COVID-19 disease was 94.1%, with
efficacy against severe COVID-19 observed to be 100%.
• Efficacy estimates were consistent across sensitivity and subgroup
analyses
• The safety profile of Moderna COVID-19 vaccine was observed to be
clinically-acceptable, and safety follow-up continues in clinical trials and
in post-authorization active and passive surveillance
• Moderna COVID-19 vaccine has the potential to be an important tool in
addressing the COVID-19 public health crisis
___________________________________________________________________
2nd Genetic Vaccine Development for Infectious Diseases
Summit:
Jakob Crammer
Head of Clinical
Development
CEPI
11.30 Clinical and Operational Challenges of COVID-19 Vaccine
Development and Future Opportunities
• Vaccine efficacy against disease versus infection / transmission: Defining
clinical trial endpoints for a new pathogen with limited pre-existing
evidence
• Establishing vaccine efficacy when placebo-controlled trials become
more and more difficult as vaccines are being licensed and deployed
• Need for correlates of protection, newly emerging variants and vaccine
adaptation, and vaccine safety and pharmacovigilance
___________________________________________________________________
Kate Broderick
Senior VP, R&D
INOVIO
12.00 INO-4800 – A DNA based Vaccine Against COVID-19
• Demonstration of efficacy in animal challenge models
• Understanding phase 1 immunogenicity and safety profile
• Proof of thermo stability of DNA vaccines and enhanced delivery through
electroporation
___________________________________________________________________
Lidia Oostvogels
VP Area Head
Infectious
Diseases
CureVac
12.30 CVnCov: CureVac’s mRNA-based Vaccine Candidate Against
COVID-19
• Discovery of targeted mRNA optimization to allow immune response
induction at low mRNA doses (incl. Rabies CV7202)
• Demonstration of clinical data to show an antibody response
comparable to natural COVID-19 infection
___________________________________________________________________
Mohadeseh
Hasanpourghadi
Post-Doctoral
Fellow
The WISTAR
Institute
2.00 Immunogenicity of Chimpanzee Adenovirus Vector Vaccines
Expressing SARS-CoV2 Spike and Nucleocapsid
• Two serologically distinct adenovirus vectors ensure optimal immune
responses in a prime boost regimen
• The AdC-Spike vaccine induces potent antibody responses to blunt
infections
• The AdC-NCap vaccine induces CD8+ T cells which may facilitate viral
clearance
___________________________________________________________________
Luk Vandenberghe
Grousbeck Associate
Professor in Gene
Therapy, Mass Eye
and Ear
Harvard Medical
School
2.30 Development of AAVCOVID: A Single Dose AAV-based Vaccine
for COVID-19
• Understanding how AAVCOVID induces potent neutralizing SARS-CoV-2
antibodies following a single dose intramuscular injection
• Developing insight into how AAVCOVID can be manufactured at scale
with established industry AAV production processes
• The AAVCOVID vaccine has minimal cold-chain requirements, with
stability at room temperature for at least 1 month
___________________________________________________________________
Amesh Adalja
Senior Scholar
John Hopkins
Centre for Health
Security
3.00 A Framework to Analyse the Pandemic Potential of Pathogens:
COVID-19 and Future Directions
• How will the integration of data form new technologies have impact on
human resilience to GCBR-level threats?
• How can we incentivise R&D for emerging pathogens, and which
pathogens with pandemic potential should we be already looking to
equip ourselves against?
• What are the biggest learning’s from the COVID-19 pandemic, and how
can we best prepare for the next?
___________________________________________________________________
Chair’s Opening Remarks
8.30 Vaccine Leader’s Panel Discussion: Optimising Next-Generation Vaccine Development,
Lessons Learned From the Pandemic, and Future Outlook
• What have been the biggest learning curves after the most recent pandemic, and how will this inform
future vaccine development?
• How can new vaccine technologies stave off future outbreaks, and what are the technologies of the
future that will help us?
• Has vaccine development changed forever, and will we continue to invest in classical vaccine platforms?
• Where will biotech and pharma look to next after COVID-19, and do we expect to continue to see
new investments and partnerships surrounding genetic vaccines?
____________________________________________________________________________
____________________________________________________________________________
Moderator:
Norman Begg
Independent
Consultant
& Former
Chief Medical
Officer
GSK Vaccines
____________________________________________________________________________
Panelists:
Johan
Vekemans
Global Clinical
Head, SARSCoV-
2 Vaccine
AstraZeneca
____________________________________________________________________________
Philip
Dormitzer
Vice President
& Chief
Scientific
Officer: Viral
Vaccines
Pfizer
____________________________________________________________________________
Andreas Kuhn
Sr. VP, RNA
Biochemistry
&
Manufacturing
BioNTech
____________________________________________________________________________
Jacqueline
Miller
Sr. VP
Infectious
Disease
Development
Moderna
____________________________________________________________________________
Introduction to Genetic Vaccinology
Norman Begg
Independent
Consultant & Former
Chief Medical Officer
GSK Vaccines
____________________________________________________________________________
Overview of Today’s Vaccine Market Highlighting Significant
Investments, Partnerships, and Opportunities
• Understanding the changing landscape of vaccine development to
identify future areas of innovation and collaboration
• How as an industry we can move to usher in a new era of vaccinology,
and what future vaccine technologies are on horizon
____________________________________________________________________________
Addressing Technical and Manufacturing Challenges to Help Accelerate Next-
Generation Vaccine Development
• What are they challenges in the development optimization of genetic vaccines?
• How is Thermo Fisher working to address these key challenges?
• What are the key learnings from the pandemic?
____________________________________________________________________________
Moderator:
Adam Taich
Vice President
& General
Manager,
Molecular
Biology
Thermo Fisher
Scientific
____________________________________________________________________________
Panellists:
Linas Pupelis
Senior
Product
Manager for
Nucleic Acid
Therapeutics
Thermo Fisher
Scientific
____________________________________________________________________________
Scott Zobbi
Business
Lead for
Custom Poros
Products
Thermo Fisher
Scientific
____________________________________________________________________________
Mathieu
Boxus
Head of R&D
for Viral Vector
Services
Thermo Fisher
Scientific
____________________________________________________________________________
Vincenza
Pironti
Subject
Matter Expert,
Sterile Drug
Products
Thermo Fisher
Scientific
____________________________________________________________________________
____________________________________________________________________________
Galit Alter
Professor of Medicine
Harvard Medical
School
12.30 Systems Serology to Define Correlates of Immunity to SARS-CoV-2
• While neutralization has been linked to protection against other
pathogens, whether neutralization alone will be sufficient to drive
protection against SARS-CoV-2 in the broader population remains unclear
• We have dissected the early evolution of the humoral response in a
cohort of hospitalized individuals ranging from moderate-to severe
• These data highlight distinct humoral trajectories associated with
resolution of SARS-CoV-2 infection and the need for early functional
humoral immunity
___________________________________________________________________
Russell Johnson
US Head Formulation
Discovery Science
GSK Vaccines
Development of a Potent Zika Virus Vaccine Using Selfamplifying
RNA
• Development of ZIKV vaccine candidates using self-amplifying mRNA
platform technology delivered by cationic nanoemulsion
• Induction of potent neutralizing antibody responses to ZIKV in mice and
non-human primates, leading to rapid protective immunity
(This note is just to put an eye on the technology of self-amplifying RNA or if you will self-replicating RNA)
___________________________________________________________________
Ami Patel
Research Assistant
Professor
The WISTAR
Institute
Optimizing DNA Vaccines Against Emerging Infectious Diseases
• Understanding DNA vaccine design and insight into the mechanism of action
• Demonstration of DNA vaccine preclinical studies, showing
immunogenicity and protection in animal models
• Understanding what constitutes next-generation DNA vaccines and
future outlook
___________________________________________________________________
Ushering in a New Era of Vaccinology: mRNA Vaccines
Susanne Rauch
Principal Scientist
CureVa
10.45 An Optimized mRNA Vaccine Platform: Broad Application
Against Various Viruses
• Analyse the use of unmodified, natural mRNA to allow for improved
induction of viral defences, including interferon type 1
___________________________________________________________________
Stefanie Krumm
Associate Director,
Infectious Disease
Vaccines
BioNTech
11.15 Development of Synthetic mRNA-based Vaccines Against
Infectious Diseases Pathogens
• Overview of BioNTech’s RNA vaccine platforms and understanding the
use of an RNA-based platform for vaccine design
• Pre-clinical evaluation using viral model systems and demonstration of
animal model data
___________________________________________________________________
Aleš Štrancar
CEO
BIA Separations
11.45 Efficient, robust and scalable purification of Covid vaccines
based on pDNA, mRNA, Adeno and VLP technologies
___________________________________________________________________
Sean Sullivan
Executive Director,
Process Development
Arcturus
Therapeutics
1.30
ARCT-021, a low dose Self-Replicating RNA based SARS-CoV-2
Prophylactic Vaccine Yielding Effective Protective Immunity
• Review of preclinical immunogenicity and virus challenge results in
transgenic mice and rhesus macaques
• Review of clinical safety and immunogenicity results
___________________________________________________________________
Self-replicating RNAs and RNA-based Delivery Methods
Luis Brito
Sr. Director,
Formulation Science
Moderna
2.00
Optimization of Lipid Nanoparticles for the Delivery of mRNA
Vaccines
• Identification of novel lipids to improve vaccine potency and tolerability
• Tailoring vaccine biophysical parameters for optimum immune responses
___________________________________________________________________
Ronit Satchi-
Fainaro
Head, Cancer
Research and
Nanomedicine
Laboratory
Tel Aviv University
2.30
Novel Approaches to Delivering miRNA and siRNAs to
Endothelial Cells: From Cancer Immunotherapy to COVID-19
Vaccine
• Establishment of preclinical models in order to rationally design novel
clinically translatable nanomedicines
• Translation of miRNA and siRNA delivery in the context of cancer to
vaccine development
___________________________________________________________________
Jesse Erasmus
Director of Virology
HDT Bio
3.00
Replicating RNA as a Platform for Active or Passive
Immunization Against Emerging Infectious Diseases
• Vaccine development activities for active immunization against
COVID-19
• Enabling intramuscular administration of RNA-encoded antibodies for
passive immunization strategies.
____________________________________________________________________________
____________________________________________________________________________
Quote;
The vaccine landscape has been revolutionised by the
advent of genetic vaccines. I am delighted to participate in this summit, to better understand how these powerful new tools can contribute to global public health.
Norman Begg, Chief Medical Officer Emeritus, GSK Vaccines
____________________________________________________________________________
________________________________________________________________________
Title: 2nd Anual Genetic Vaccine Development for Infectious Diseases Summit
(Powering the Next Generation of Gene-Based Vaccines)
____________________________________________________________________________
Slogan:
Dedicated to Delivering the New Era of Vaccinology Through Optimizing
the Clinical Translation, Discovery, & Process Development of mRNA, DNA,
& Viral Vector-Based Vaccines for Respiratory & Infectious Diseases
Worldwide
____________________________________________________________________________
____________________________________________________________________________
Speakers:
Joshua DiNapoli
Global Project Head,
mRNA Platform
Development
Sanofi Pasteur
____________________________________________________________________________
Kay Hunsberger
Director of
Infrastructure for
Biological Clinical
Manufacturing
Merck
____________________________________________________________________________
Grace Chen
Senior Director, Clinical
Development, Infectious
Disease
Moderna
____________________________________________________________________________
Dushyant Varshney
Chief Technology
Officer
Arcturus Therapeutics
____________________________________________________________________________
David Morrissey
Vice President & Head,
RNA Accelerator
Pfizer
________________________________________________________________________________________________________________________________________________________
Andrew Geall
Chief Development
Officer
Replicate Bio
____________________________________________________________________________
Martin Friede
Coordinator
Initiative for Vaccine
Research, World Health
Organization
____________________________________________________________________________
____________________________________________________________________________
Intro:
REUNITE IN-PERSON WITH THE VACCINE COMMUNITY
Overcome the Translational Challenges of Developing mRNA,
DNA & Viral Vector-Based Vaccines for Infectious Diseases
____________________________________________________________________________
It has been a landmark year for gene-based vaccines once again, with the development of the first mRNA COVID-19 booster doses, approval of the world’s first
DNA-based vaccine for human use, and the continued clinical development of viral vector-based vaccines.
The 2nd Genetic Vaccine Development for Infectious Diseases Summit (GVID) will
reunite the vaccine community to learn from innovative biotechs, trailblazing large pharma, and pioneering academics as we focus on overcoming the pre-clinical and
clinical translational challenges of developing genetic vaccines targeting infectious diseases. With a larger focus on respiratory infectious diseases, GVID provides
end-to-end detailed insight into the development of next-generation vaccine candidates for COVID-19 and beyond.
This summit is built specifically for those looking to integrate novel vaccine development approaches into their pre-clinical studies through to clinical translation, covering mRNA, srRNA, pDNA, adenovirus, RV, and AAV-based
vaccine platforms.
____________________________________________________________________________
Join 80+ senior decision-makers from pre-clinical and clinical development, infectious diseases, virology and R&D from the world’s
leading vaccine developers while accessing ground-breaking content, and hours of networking, as we deliver the next era of vaccinology to prevent infectious diseases
worldwide.
____________________________________________________________________________
DISCOVERY
Starting with the science, discover the pioneering platforms being implemented across the pharma
and biotech value chain, such as how HDT Bio is implementing RNA replicating technology as a platform for active and passive immunization, and how The
WISTAR Institute are developing several viral-vector-based vaccines against multiple infectious diseases.
____________________________________________________________________________
MANUFACTURE
Ensuring a GMP-certified manufacturing process is
fundamental for the successful scale-up and production of vaccines.
Understand how to achieve efficient, robust, and scalable purification of COVID-19 vaccines based on pDNA, mRNA, and Adeno technologies from BIA Separations & Precision NanoSystems.
____________________________________________________________________________
PRECLINICAL
Discussing early vaccine candidates showing promise for
the future and learning from previous vaccine studies to
inform current candidates post-discovery whilst developing
the right pre-clinical models to assess immunogenicity
profiles from Moderna, Arcturus Therapeutics & Sanofi.
________________________________________________________________________________________________________________________________________________________
CLINICAL
With an increasing number of next-generation vaccine
candidates entering the clinic, understand the clinical and
operational challenges of COVID-19 vaccine development from CEPI and learn how clinical work carried out during the Ebola outbreak informed rapid clinical translation of a COVID-19 vaccine from Merck.
____________________________________________________________________________
____________________________________________________________________________
Qoute;
The vaccine landscape has been revolutionised by the advent of genetic vaccines. I am delighted to participate in this summit, to better understand how these powerful new tools can contribute to global public health.
Norman Begg, Chief Medical Officer Emeritus, GSK Vaccines
____________________________________________________________________________
2 Tel: (+1) 616 455 4188 Email: info@hansonwade.com www.genetic-vaccine-development.com
___________________________________________________________________
___________________________________________________________________
Future Directions & Adapting to COVID-19
David Morrissey
Vice President
& Head, RNA
Accelerator
Pfizer
____________________________________________________________________________
8.00 Advancing Pfizer’s mRNA Strategy
• David Morrissey, Head of Pfizer’s RNA Accelerator, discusses the company’s strategy for advancing its leadership in mRNA and related
technology platforms
____________________________________________________________________________
8.30
Breakfast Panel: What does the Future Landscape of Vaccinology Look Like?
How Can we Further Leverage mRNA Technology in Vaccinology & How Can Pharma & Biotech Prepare to Expand?
• Could self-amplifying RNA be a more potent version of mRNA?
• Could mRNA be used to develop vaccines that target numerous strains of a virus family, like coronaviruses and influenza, and act as a first-line defence against future pandemics?
• What are the challenges of developing, manufacturing and scaling up these new modalities? How can we meet these challenges?
____________________________________________________________________________
Kay Hunsberger
Director of Infrastructure
for Biological Clinical
Manufacturing
Merck
____________________________________________________________________________
Denis Vargo
Vice President, Head of Drug
Safety & Pharmacovigilance
Akebia Therapeutics
____________________________________________________________________________
Joshua DiNapoli
Global Project Head, mRNA
Platform Development
Sanofi Pasteur
____________________________________________________________________________
Afam Okoye
Research Associate Professor
VGTI-Vaccine & Gene
Therapy Institute
OHSU
____________________________________________________________________________
Andrew Geall
Chief
Development
Officer
Replicate Bio
____________________________________________________________________________
____________________________________________________________________________
James Cody
Senior Manager,
Business
Development,
Biologics Solutions
Charles River Labs
____________________________________________________________________________
9.15
Overcoming Challenges in Genetic Vaccine Development – Manufacturing and Testing
• Overview of hurdles for genetic vaccine development
• Focus on key challenges involved manufacturing and testing of DNA and viral vector vaccines
• How CRL’s end-to-end support can help product developers avoid and overcome these challenges
____________________________________________________________________________
Amesh Adalja
Senior Scholar
John Hopkins
Centre
for Health Security
____________________________________________________________________________
10.45
A Framework to Analyze the Pandemic Potential of Pathogens
___________________________________________________________________
Emma Thompson
Professor of
Infectious
Diseases
Centre for
Virus Research,
University of
Glasgow
____________________________________________________________________________
11.15
SARS-CoV-2 Genetic Variation and Vaccine Effectiveness
• Tracking of the SARS-CoV-2 variant through high-throughput genomic sequencing was used in the UK as an “early warning system”
• Linked datasets have helped to associate vaccine effectiveness with genetic variation
• Phenotypic changes in new variants cannot always initially be predicted through sequencing
___________________________________________________________________
Sean Sullivan
Executive
Director
Arcturus
Therapeutics
____________________________________________________________________________
11:45
Pre-Clinical and Clinical Development of a Next-Generation mRNA COVID-19 Vaccine
• Preliminary preclinical results for second generation vaccines designed with improved anti-viral immunogenicity exhibited cross neutralizationagainst
alpha, beta, gamma and delta circulating viral variants in mice and non-human primates
• Rhesus macaque immunogenicity studies showed high neutralizing antibody titers after two prime injections 28 days apart
• A further increase in neutralizing antibody titers were observed with a boost injection 120 days after the second prime injection
___________________________________________________________________
Rok Sekirnik
Head Process
Development
mRNA/pDNA
BIA Separations
____________________________________________________________________________
12.15 Increasing Productivity of IVT Reaction Using At-Line Monitoring by HPLC Chromatography
___________________________________________________________________
Advancing Next-Generation Vaccine Process, Pre-Clinical, & Clinical Development
Ami Patel
Research
Assistant
Professor
The WISTAR
Institute
____________________________________________________________________________
1.45
Optimizing DNA Vaccines Against Emerging Infectious Diseases
• Understanding DNA vaccine design and insight into mechanism of action
• Demonstrating of DNA vaccine preclinical studies
• immunogenicity and protection in animal models
• Understanding what constitutes next generation DNA vaccines and future outlook
___________________________________________________________________
Andrew Geall
Chief
Development
Officer
Replicate Bio
____________________________________________________________________________
2.15
Next Generation Self-Replicating RNA Vectors to Enable New Approaches to Immunotherapy Synopsis
• Next generation synthetic srRNA vectors can lower dose by orders of magnitude compared to current srRNA designs
• Developing new srRNA products requires independent optimization of vectors, gene inserts, and delivery
• Improved srRNA bioactivity enable new approaches for strategies in vaccine development
___________________________________________________________________
____________________________________________________________________________
Grace Chen
Senior Director,
Clinical
Development
Moderna
2.45
Clinical Updates of mRNA-1345, Moderna’s Prophylactic Vaccine for RSV
• Learn how an mRNA vaccine against RSV encoding a pre-fusion F glycoprotein, can lead to a superior neutralizing antibody response compared to
the post-fusion state
• Phase 3 data to show promising results of RSV prevention in older adults
___________________________________________________________________
Jason Coleman
Clinical Application
Scientist Lead
Precision Nano
Systems
3.15
Scalable Manufacturing Platform for RNA-LNP Vaccine Development
• COVID-19 mRNA vaccines, while an extraordinary accomplishment, have revealed that rapid development and scale-up, as well as access to innovative technologies, are needed to keep up with emerging variants and to usher in a new wave of genomic medicines
• Challenges associated with addressing a global pandemic include the need for a scalable, quick and localized manufacturing platform as well as the need for a higher potency vaccine
• In this presentation, we will share insights into a cutting-edge technology to enable scalable manufacturing for RNA-LNPs and the use of this technology for the development of a high potency saRNA-LNP COVID vaccine
___________________________________________________________________
Doug Holtzman
Chief Scientific
Officer
ICOSAVAX
4:15
A Novel Two-Component VLP Technology for the Development of Vaccines Against Respiratory Viruses
• Vaccine technology covering an RSV/hMPV bivalent vaccine as well as a SARS2 candidate which is in Ph1 testing now in Australia
• Advantages of computational protein design to better inform antigen formulation and to achieve higher efficacy rates
___________________________________________________________________
Jesse Erasmus
Director of Virology
HDT Bio
4.45
Replicating RNA as a Platform for Active or Passive Immunization Against Emerging Infectious
• Vaccine development activities for active immunization against COVID-19
• Enabling intramuscular administration of RNA-encoded antibodies for passive immunization strategies
___________________________________________________________________
Anna Blakney
Assistant Professor
University of British
Columbia
5.15
Unravelling Viral & Non-Viral Delivery Approaches for Next Generation RNA Therapeutics
• Understanding the stability and quality differences between viral and non-viral delivery strategies
• Understanding biological mechanism of non-viral delivery strategies to improve targeted delivery
• Choosing the most appropriate delivery system for different types
____________________________________________________________________________
Hildegund Ertl
Professor
The WISTAR
Institute
5.45
Therapeutic HBV Vaccine to Target Internal Viral Proteins: Clinical Development
• Methods Vaccines are delivered by chimpanzee adenovirus vectors (AdC) of serotype 6 (AdC6) and 7 (AdC7) used in prime only or prime-boost
regimens
• Results show that the vaccines are immunogenic in mice and induce potent CD8⁺ T cell responses that recognize multiple epitopes
____________________________________________________________________________
Future Directions & Adapting to COVID-19
Kay Hunsberger
Director of
Infrastructure for
Biological Clinical
Manufacturing
Merck
8.30
Developing Vaccines at Pandemic Speed
• Highlighting approaches in vaccine development to be prepared for future potential pandemic infectious diseases and share our historical
experiences with Ebola and Coronavirus
• How Merck responded to the Ebola outbreak, share our experience partnering with other companies, government, health, and regulatory
agencies, and non-government organizations to develop and implement a vaccine to address the outbreak
• How Merck’s development efforts for our COVID-19 program were significantly accelerated by using the previously-developed Ebola platform
• Lessons learned and future preparations to respond to the next global infectious disease
___________________________________________________________________
akob Cramer
Head of Clinical
Development
CEPI
9.00
Clinical & Operational Challenges of COVID-19 Vaccine Development & Future Opportunities
• Vaccine efficacy against disease versus infection / transmission: Defining clinical trial endpoints for a new pathogen with limited
pre-existing evidence
• Establishing vaccine efficacy when placebo-controlled trials become more and more difficult as vaccines are being licensed and
deployed
• Need for correlates of protection, newly emerging variants and vaccine adaptation, and vaccine safety and pharmacovigilance
____________________________________________________________________________
Nerea Zabaleta
Lasarte
Postdoctoral
Research Fellow
Grousbeck Gene
Therapy Center
Harvard Medical
School
9.30
Clinical Development of AAVCOVID: A Single Dose AAV-Based Vaccine for COVID-19
• Understanding how AAVCOVID induces potent neutralizing SARS-CoV-2 antibodies following a single dose intramuscular injection
• Developing insight into how AAVCOVID can be manufactured at scale with established industry AAV production processes
• The AAVCOVID vaccine has minimal cold-chain requirements, with stability at room temperature for at least 1 month
____________________________________________________________________________
Martin Friede
Coordinator, Initiative
for Vaccine Research
World Health
Organization
____________________________________________________________________________
Birgitte Giersing
Technical Officer,
Initiative for Vaccine
Research
World Health
Organisation
____________________________________________________________________________
10.00
Establishment of a COVID-19 mRNA Vaccine Technology Transfer Hub to Scale Up Global Manufacturing
• Expand the capacity of low- and middle-income countries (LMICs) to produce COVID-19 vaccines and scale up
manufacturing to increase global access to these critical tools to bring the pandemic under control
• Enabling production of mRNA-based COVID-19 vaccines in LMICs through unprecedented partnerships and creation to
technology-transfer hubs
___________________________________________________________________
Galit Alter
Professor of Medicine
Harvard Medical
School
11.30
Systems Serology to Define Updated Correlates of Immunity to SARS-CoV-2
• While neutralization has been linked to protection against other pathogens, whether neutralization alone will be sufficient to drive
protection against SARS-CoV-2 in the broader population remains unclear
• Dissecting the early evolution of the humoral response in a cohort of hospitalized individuals ranging from moderate-to severe
• Highlighting distinct humoral trajectories associated with resolution of SARS-CoV-2 infection and the need for early functional humoral immunity
___________________________________________________________________
Don Diamond
Professor, Cancer and
Viral Vaccines
City of Hope
12.00
Conception, Construction, Pre-Clinical & Clinical Testing of COH04S1 Vaccine with Activity Against SARS-CoV-2
• COVID-19 variants have arisen with different biologic properties culminating in the most recent and frightening variety referred to as
Omicron. Development of a vaccine that would have prophylactic activity against a variety of SARS-CoV-2 strains by expanding the antigen cargo two include both the Nucleocapsid and Spike antigens
• Describing the development process of a vaccine including a proprietary synthetic viral platform, laboratory and animal studies,
culminating with an overview of ongoing and completed Phase 1 and Phase 2 trials in healthy adults and immunocompromised
cancer patients and stem cell transplant recipients
• Sharing results from Phase 1b trial (NCT046339466) and ongoing Phase 2 trials (NCT04977024) to demonstrate the capacity of a
vaccine to stimulate protective humoral and cellular immunity in healthy persons and cancer patients
___________________________________________________________________
Simon van Haren
Research Associate,
Division of Infectious
Diseases, Precision
Vaccines Program
Boston Children’s
Hospital
12.30
Exploring Vaccine Adjuvant R&D Within RSV Models
• Preclinical studies showing effectiveness in neonates
• Clinical development of the CAF®01 adjuvant
• Immune profile of correct Th1/Th2 and antibody responses for optimal protection against RSV
____________________________________________________________________________
Sanie Sesay
Global Director
Clinical Development
Vaccines Sanofi
Pasteur
2.00
Developing a Live Attenuated RSV Vaccine for Infants Newborns & Toddlers
• Recent advances in the understanding of RSV gene function and reverse genetics have led to the rational design of promising new-generation
replication-competent live attenuated RSV vaccines
• Live attenuated RSV vaccines do not cause enhanced disease
• Live attenuated RSV vaccines are expected to induce innate immunity, durable local and systemic immunity
• Development of a live attenuated RSV vaccine could substantially reduce the global burden of disease in infants and toddlers
____________________________________________________________________________
Geeta Swamy
Associate Vice
President for
Research Vice Dean
for Scientific Integrity
Professor, Obstetrics
& Gynecology
Duke University
2.30
Observational Maternal COVID-19 Vaccination Clinical Data
• Injection-site (local) and systemic reaction data will be discussed and second vaccination
• Analyzing maternal serum samples to compare for antibody titers relevant to COVID-19 at time points that include: prior to vaccination, ~29 days
post second vaccination, and at delivery
• Maternal and infant cord blood analyzed for the same antibody titers
____________________________________________________________________________
Dushyant Varshney
Chief Technology
Officer
Arcturus
Therapeutics
3.00
Global Technology Transfer & Commercialization of Next-Gen mRNA Vaccines
____________________________________________________________________________
Paes
Professor Emeritus,
Pediatrics
McMaster University
4.00
The Canadian Registry of Palivizumab (CARESS): Outcomes Following Twelve Seasons of RSV Prophylaxis
• Review the CARESS study protocol and enrolment
• Examine the indications for prophylaxis, palivizumab utilization and adherence
• Review the respiratory and RSV-related hospitalizations across the indications from 2005-2017
____________________________________________________________________________
Leonard Krilov
Chairman,
Department of
Pediatrics
NYU Long Island
School of Medicine
4.30
Updated RSV Epidemiology of High-Risk Infants & Toddlers
• Updated overview of the morbidity in developed countries and in high-risk neonates and potential implications
• Potential targeted prevention of RSV infection based on current epidemiology
• Short term burden of RSV infection
____________________________________________________________________________
Ian Mitchell
Professor Emeritus,
Department of
Pediatrics
University of
Calgary
5.00
The Burden of RSV Infection on Healthcare Systems, Children & Families
• RSV infection remains most common cause of respiratory admissions in infancy
• Abrupt onset of RSV epidemics creates annual resource problems
• Effective, safe RSV vaccine immediate system impact
____________________________________________________________________________
PARTNER WITH US SNAPSHOT OF PREVIOUS ATTENDEES
Partnering with the 2nd Genetic Vaccine Development for Infectious Diseases, Summit is your opportunity to demonstrate your expertise, elevate your brand, and
establish yourself as a key thought leader and front runner within the vaccine community. Your brand, your message, and your reputation showcased in front
of translational and clinical experts from the field of vaccine development.
With a huge amount of excitement and innovation within the vaccines space at the moment, and an ever-increasing number of companies launching new
vaccine candidates into pre-clinical and clinical studies, there is a unique opportunity for pre-clinical CROs, viral-vector providers, adjuvant formulators, and immune
reagent suppliers to establish themselves as the go to commercial partners for the industry. We will work with you to build bespoke partnerships and ensure you meet your 2022 business objectives. Get intouch today to learn more about how we can support you and your commercial goals within the vaccine development field.
____________________________________________________________________________
George Shrimpton
Senior Partnerships Director
E: Sponsor@hansonwade.com
T: +1 617 455 4188
____________________________________________________________________________
____________________________________________________________________________
From A Book On Gene Therapy;
CRISPR (clustered regularly interspaced short palindromic repeats) sequences and Cas (CRISPR-associated) proteins are a biological system for permanent genome editing. The CRISPR/Cas system consists of a recombinant Cas (typically Cas9 or CPF1) enzyme that catalyzes double-stranded DNA cleavages with extraordinary specificity based on hybridization of the synthetic CRISPR targeting sequence (sgRNA) against the target genomic DNA. Furthermore, HDR edited or corrected DNA sequences can be inserted into the genome.
As previously mentioned, the CRISPR/ Cas system is a combination of both synthetic and recombinant components. Each component needs to be independently manufactured and then formulated together. An emerging approach is to combine both a guide RNA sequence and Cas mRNA protein sequence into a single plasmid. Nonetheless, both the single plasmid and plasmid/ RNA combination require either formulation into lipid nanoparticles or packaging into a viral vector for delivery.
An additional challenge demonstrated by investigational CRISPR/Cas therapies is the need to control dose and/or nuclear residence time to carefully balance efficacy with off-target effects.
____________________________________________________________________________
____________________________________________________________________________
SCIEX Gene Therapy and Oligonucleotide Compendium
Introduction
Currently, there are hundreds of ongoing clinical trials for various gene therapies ranging from inherited genetic disorders to cancer and viral infections and even heart disease and diabetes. With the potential to prevent, treat or even cure many types of diseases, gene therapy-related research and development continues to be a highly active and growing area within pharmaceutical companies.
This past year has been particularly challenging for many pharmaceutical companies as the novel SARS-CoV-2 virus swept the world in a deadly pandemic not seen since the emergence of H1N1 influenza (Spanish flu) over a century before.
Many pharmaceutical programs were suspended as global scientific communities raced to develop and test vaccines for the virus. The dramatic all-hands-on- deck efforts of governments and vaccine developers working in tandem resulted in the unprecedented rapid emergency approval of safe and effective vaccines, including the first-ever RNA-based genetic vaccines, in less than a year.
Although they are not considered gene therapy, genetic vaccines do share common active ingredients and delivery systems with gene therapeutics. The synthesis of RNA- or DNA-based active agents and their packaging within lipid nanoparticles (LNP) or adeno-associated viral (AAV) vectors requires many of the same analytical tools that are used for the design, creation and purification of gene therapies. As a result, the development of the SARS-CoV-2 vaccines has not only helped to combat a serious public health crisis in the immediate term but also laid the groundwork for improvements going forward. The tools and infrastructures used in this process will help to streamline future vaccine programs while facilitating the development of gene therapies within biopharmaceutical pipelines.
In the early development of gene therapies, scientists borrowed techniques from the biotherapeutic development process. For example, capillary electrophoresis sodium dodecyl sulfate (CE-SDS) is a technique adopted from biologics development that has proven to be highly beneficial for AAV capsid protein purity analysis. In contrast, many existing technologies from biotherapeutics development have limitations when characterizing gene therapeutics. As a result, new strategies have been designed to meet the unique challenges of gene therapy development, such as using capillary isoelectric focusing (cIEF) as a simple and robust method for the characterization of AAV capsid protein contents.
____________________________________________________________________________
SCIEX Gene Therapy and Oligonucleotide Compendium
____________________________________________________________________________
____________________________________________________________________________
The evolution of gene therapy
Mani Krishnan
Vice President and General Manager, CE and Biopharma Business Unit, SCIEX
____________________________________________________________________________
While the promise and investment in the development of gene therapies are high, the technologies for the manufacturing and analysis of gene-therapy products continue to evolve.
Gene therapy is a concept that is over 40 years old. Although there were setbacks in the 1990s, many of the challenges that have hindered progress are now resolved and mitigated. Since then, there has been a massive resurgence in the development of gene therapeutics, with over 1,000 clinical trials that are either ongoing or completed. While most of these trials are for therapies that address single-mutation diseases, there are multigene diseases that may be cured by gene therapies. In fact, more than 7,000 genetic diseases could potentially be cured using gene therapy, so the promise is substantial.
The current state of gene-therapy development and manufacturing is similar to the early days of protein therapeutics development and production. There is an expectation that we will follow a model in maturing gene-therapy production that is analogous to protein therapeutics but moves at a much faster pace. However, there are some fundamental differences between the production of protein therapies and gene therapies that present new and complex challenges. With protein therapies, the DNA sequence for the protein therapeutic is introduced into the host cell line.
Cells that produce the highest quality and quantity of protein products become the master cell bank and are extensively characterized and scaled up for therapeutic protein production. While there may be minor modifications within the pool of proteins that are produced, this well-characterized master cell bank is enormously beneficial for ensuring that a reliable and well-characterized product can be manufactured. With gene therapy, a viral vector such as an associated virus (AAV) is often used for the delivery of the gene product to the patient. Viral vector production requires the production cell line to be transfected by a series of plasmid complexes. The cells must then make the necessary components and correctly assemble the AAV particles.
The pool of recombinant AAV is then characterized and purified. There is no master cell bank and every individual host cell requires plasmid transfection for viral-vector production. The need for each cell to be transfected by plasmid complexes increases the probability of inducing product heterogeneity. Elimination of the transfection process to produce a stable transfected cell line can remove a lot of variability in the entire process. Producing stable transfected cell So, for now, most products in the clinic or in the market today still use transient transfection for producing viral modalities.
____________________________________________________________________________
SCIEX Gene Therapy and Oligonucleotide Compendium
____________________________________________________________________________
Viral gene therapies are recombinantly produced systems composed of a genetic construct consisting of essential viral genes, a therapeutic gene (transgene) to be introduced and associated promoters, all contained within a viral capsid.
Although there are many types of viral vector systems, by far the 2 most commonly used are adeno-associated virus (AAV) and lentivirus (LV). Viral gene therapies offer the capability of introducing extensive amounts of genetic instructions to a wide variety of target cell types with high efficiency to produce nearly any type of protein. Unfortunately, viral gene therapies are very difficult to produce in high yields with high purity. Viral vector biosynthesis typically requires multiple plasmid transfections to HEK293 cells with varying efficiencies and outcomes with each transfection step. Timely and accurate analysis of transfection steps and viral production will lower the cost as well as improve the safety of these therapeutics.
Therapeutic oligonucleotides are chemically synthesized strands of nucleotides in the range of 15-30 bases. Therapeutic oligonucleotides are broadly broken down into 2 classes: antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs). Whether ASO or siRNA, these oligonucleotide sequences are highly chemically modified to increase both plasma and intracellular stability. Both classes selectively target the mRNA transcript of the protein to be blocked, utilizing
either RNAseH for ASOs or RISC for siRNAs or the pre-mRNA transcript of the protein to be modulated via differential splicing. Therapeutic oligonucleotides are difficult to produce in high yields with high purity, and they are difficult to formulate for efficient delivery and stability. Furthermore, oligonucleotides have a degree of regulatory uncertainty since their biophysical characteristics straddle the line between small molecules and biologics.
____________________________________________________________________________
SCIEX Gene Therapy and Oligonucleotide Compendium
____________________________________________________________________________
____________________________________________________________________________
From the Pfizer purchase agreement with the EU.
Document name;
APA BioNTech Pfizer - ADVANCE PURCHASE AGREEMENT.pdf
(Unredacted version)
____________________________________________________________________________
____________________________________________________________________________
The first reply I got from the Head of Health on the EU Commission;
________________________________________________________________________
The second reply I got from the Head of Health on the EU Commission;
Damage report from Eu database:
44,821 Dead 4,351,483 Injured Following COVID-19 Vaccines in European Database of Adverse Reactions:
Here is the summary data through May 21, 2022.
Total reactions for the mRNA vaccine Tozinameran (code BNT162b2, Comirnaty) from BioNTech/ Pfizer: 21,070 deaths and 2,207,919 injuries to 21/05/2022
67,512 Blood and lymphatic system disorders incl. 303 deaths
78,229 Cardiac disorders incl. 3,061 deaths
694 Congenital, familial and genetic disorders incl. 63 deaths
29,164 Ear and labyrinth disorders incl. 16 deaths
2,801 Endocrine disorders incl. 10 deaths
32,801 Eye disorders incl. 54 deaths
161,894 Gastrointestinal disorders incl. 837 deaths
550,894 General disorders and administration site conditions incl. 5,821 deaths
2,542 Hepatobiliary disorders incl. 121 deaths
24,583 Immune system disorders incl. 134 deaths
123,754 Infections and infestations incl. 2,182 deaths
41,268 Injury, poisoning and procedural complications incl. 441 deaths
53,565 Investigations incl. 582 deaths
14,151 Metabolism and nutrition disorders incl. 368 deaths
253,409 Musculoskeletal and connective tissue disorders incl. 287 deaths
2,466 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 241 deaths
348,167 Nervous system disorders incl. 2,266 deaths
3,188 Pregnancy, puerperium and perinatal conditions incl. 85 deaths
301 Product issues incl. 4 deaths
39,004 Psychiatric disorders incl. 242 deaths
8,112 Renal and urinary disorders incl. 312 deaths
95,775 Reproductive system and breast disorders incl. 8 deaths
92,246 Respiratory, thoracic and mediastinal disorders incl. 2,233 deaths
98,876 Skin and subcutaneous tissue disorders incl. 185 deaths
5,415 Social circumstances incl. 27 deaths
25,013 Surgical and medical procedures incl. 251 deaths
52,095 Vascular disorders incl. 936 deaths
________________________________________________________________________
Total reactions for the mRNA vaccine SPIKEVAX/mRNA-1273 (CX-024414) from Moderna: 11,916 deaths and 720,662 injuries to 21/05/2022
19,456 Blood and lymphatic system disorders incl. 134 deaths
24,903 Cardiac disorders incl. 1,315 deaths
224 Congenital, familial and genetic disorders incl. 15 deaths
8,515 Ear and labyrinth disorders incl. 8 deaths
741 Endocrine disorders incl. 6 deaths
9,632 Eye disorders incl. 36 deaths
57,258 Gastrointestinal disorders incl. 451 deaths
190,630 General disorders and administration site conditions incl. 3,850 deaths
968 Hepatobiliary disorders incl. 64 deaths
8,062 Immune system disorders incl. 39 deaths
31,759 Infections and infestations incl. 1089 deaths
10,916 Injury, poisoning and procedural complications incl. 215 deaths
15,827 Investigations incl. 405 deaths
5,813 Metabolism and nutrition disorders incl. 282 deaths
89,472 Musculoskeletal and connective tissue disorders incl. 238 deaths
860 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 92 deaths
118,849 Nervous system disorders incl. 1,146 deaths
1,032 Pregnancy, puerperium and perinatal conditions incl. 13 deaths
120 Product issues incl. 3 deaths
11,834 Psychiatric disorders incl. 193 deaths
3,818 Renal and urinary disorders incl. 230 deaths
19,671 Reproductive system and breast disorders incl. 9 deaths
29,456 Respiratory, thoracic and mediastinal disorders incl. 1,264 deaths
37,258 Skin and subcutaneous tissue disorders incl. 107 deaths
2,747 Social circumstances incl. 45 deaths
5,716 Surgical and medical procedures incl. 218 deaths
15,125 Vascular disorders incl. 449 deaths
________________________________________________________________________
Total reactions for the vaccine AZD1222/VAXZEVRIA (CHADOX1 NCOV-19) from Oxford/ AstraZeneca: 8,958 deaths and 1,267,883 injuries to 21/05/2022
14,856 Blood and lymphatic system disorders incl. 320 deaths
23,037 Cardiac disorders incl. 946 deaths
269 Congenital familial and genetic disorders incl. 10 deaths
14,620 Ear and labyrinth disorders incl. 7 deaths
784 Endocrine disorders incl. 6 deaths
21,601 Eye disorders incl. 33 deaths
114,466 Gastrointestinal disorders incl. 474 deaths
337,830 General disorders and administration site conditions incl. 2,084 deaths
1,154 Hepatobiliary disorders incl. 80 deaths
6,407 Immune system disorders incl. 44 deaths
54,461 Infections and infestations incl. 745 deaths
14,617 Injury poisoning and procedural complications incl. 219 deaths
30,652 Investigations incl. 223 deaths
14,191 Metabolism and nutrition disorders incl. 142 deaths
182,458 Musculoskeletal and connective tissue disorders incl. 188 deaths
854 Neoplasms benign malignant and unspecified (incl cysts and polyps) incl. 50 deaths
252,545 Nervous system disorders incl. 1,298 deaths
698 Pregnancy puerperium and perinatal conditions incl. 22 deaths
205 Product issues incl. 1 death
22,791 Psychiatric disorders incl. 78 deaths
4,703 Renal and urinary disorders incl. 91 deaths
18,099 Reproductive system and breast disorders incl. 3 deaths
44,746 Respiratory thoracic and mediastinal disorders incl. 1,200 deaths
56,079 Skin and subcutaneous tissue disorders incl. 77 deaths
2,221 Social circumstances incl. 10 deaths
2,869 Surgical and medical procedures incl. 39 deaths
30,670 Vascular disorders incl. 568 deaths
________________________________________________________________________
Total reactions for the COVID-19 vaccine JANSSEN (AD26.COV2.S) from Johnson & Johnson: 2,877 deaths and 152,918 injuries to 21/05/2022
1,529 Blood and lymphatic system disorders incl. 65 deaths
3,353 Cardiac disorders incl. 244 deaths
55 Congenital, familial and genetic disorders incl. 2 deaths
1,688 Ear and labyrinth disorders incl. 3 deaths
135 Endocrine disorders incl. 2 deaths
2,012 Eye disorders incl. 12 deaths
10,769 Gastrointestinal disorders incl. 106 deaths
41,462 General disorders and administration site conditions incl. 816 deaths
191 Hepatobiliary disorders incl. 17 deaths
713 Immune system disorders incl. 11 deaths
12,281 Infections and infestations incl. 246 deaths
1,425 Injury, poisoning and procedural complications incl. 31 deaths
7,093 Investigations incl. 154 deaths
950 Metabolism and nutrition disorders incl. 77 deaths
19,497 Musculoskeletal and connective tissue disorders incl. 63 deaths
123 Neoplasms benign, malignant and unspecified (incl cysts and polyps) incl. 11 deaths
26,795 Nervous system disorders incl. 274 deaths
110 Pregnancy, puerperium and perinatal conditions incl. 1 death
34 Product issues
2,205 Psychiatric disorders incl. 31 deaths
666 Renal and urinary disorders incl. 43 deaths
3,864 Reproductive system and breast disorders incl. 6 deaths
5,401 Respiratory, thoracic and mediastinal disorders incl. 358 deaths
4,636 Skin and subcutaneous tissue disorders incl. 13 deaths
546 Social circumstances incl. 7 deaths
1,119 Surgical and medical procedures incl. 96 deaths
4,266 Vascular disorders incl. 188 deaths
________________________________________________________________________
Total reactions for the COVID-19 vaccine NUVAXOVID (NVX-COV2373) from Novavax: 0 deaths and 2,101 injuries to 21/05/2022
44 Blood and lymphatic system disorders
116 Cardiac disorders
26 Ear and labyrinth disorders
1 Endocrine disorders
46 Eye disorders
154 Gastrointestinal disorders
463 General disorders and administration site conditions
3 Hepatobiliary disorders
8 Immune system disorders
102 Infections and infestations
19 Injury, poisoning and procedural complications
57 Investigations
13 Metabolism and nutrition disorders
275 Musculoskeletal and connective tissue disorders
2 Neoplasms benign, malignant and unspecified (incl cysts and polyps)
373 Nervous system disorders
1 Pregnancy, puerperium and perinatal conditions
1 Product issues
36 Psychiatric disorders
11 Renal and urinary disorders
39 Reproductive system and breast disorders
105 Respiratory, thoracic and mediastinal disorders
142 Skin and subcutaneous tissue disorders
3 Social circumstances
9 Surgical and medical procedures
52 Vascular disorders
________________________________________________________________________
Awareness;
________________________________________________________________________
Notes from;
Gene_Therapy_Next_Generation_Vector_Summit_brochure.pdf
________________________________________________________________________
Covid-19 Survival rate before experimental Gene Therapy Injections;
________________________________________________________________________
A document that was said to be inaccurate;
Link to document:
https://www.fda.gov/media/143557/download
In text form;
___________________________________
Guillain-Barré syndrome
___________________________________
Acute disseminated encephalomyelitis
___________________________________
Transverse myelitis
___________________________________
Encephalitis/ myelitis/ encephalomyelitis /meningoencephalitis/ meningitis/ encepholapathy
___________________________________
Convulsions/seizures
___________________________________
Stroke
___________________________________
Narcolepsy and cataplexy
___________________________________
Anaphylaxis
___________________________________
Acute myocardial infarction
___________________________________
Myocarditis/pericarditis
___________________________________
Autoimmune disease
___________________________________
Deaths
___________________________________
Pregnancy and birth outcomes
___________________________________
Other acute demyelinating diseases
___________________________________
Non-anaphylactic allergic reactions
___________________________________
Thrombocytopenia
___________________________________
Disseminated intravascular coagulation
___________________________________
Venous thromboembolism
___________________________________
Arthritis and arthralgia/joint pain
___________________________________
Kawasaki disease
___________________________________
Multisystem Inflammatory Syndrome in Children
___________________________________
Vaccine enhanced disease
________________________________________________________________________
Pfizer Document:
5.3.6 CUMULATIVE ANALYSIS OF POST-AUTHORIZATION ADVERSE EVENT
REPORTS OF PF-07302048 (BNT162B2) RECEIVED THROUGH 28-FEB-2021
9 Pages of undisclosed potential adverse reactions.
See;
APPENDIX 1. LIST OF ADVERSE EVENTS OF SPECIAL INTEREST
________________________________________________________________________
APPENDIX 1. LIST OF ADVERSE EVENTS OF SPECIAL INTEREST
________________________________________________________________________
Over 1100 Potential reactions.
Page 1;
________________________________________________________________________
1p36 deletion syndrome;
2-Hydroxyglutaric aciduria;
5'nucleotidase increased;
Acoustic neuritis;
Acquired C1 inhibitor deficiency;
Acquired epidermolysis bullosa;
Acquired epileptic
aphasia;Acute cutaneous lupus erythematosus;
Acute disseminated encephalomyelitis;
Acute encephalitis with refractory,
repetitive partial seizures;
Acute febrile neutrophilic dermatosis;
Acute flaccid myelitis;
Acute haemorrhagic leukoencephalitis;
Acute haemorrhagic oedema of infancy;
Acute kidney injury;
Acute macular outer retinopathy;
Acute motor axonal neuropathy;
Acute motor-sensory axonal neuropathy;
Acute myocardial infarction;
Acute respiratory distress syndrome;
Acute respiratory failure;Addison's disease;
Administration site thrombosis;
Administration site vasculitis;
Adrenal thrombosis;
Adverse event following immunisation;
Ageusia;Agranulocytosis;Air embolism;
Alanine aminotransferase abnormal;
Alanine aminotransferase increased;
Alcoholic seizure;
Allergic bronchopulmonary mycosis;
Allergic oedema;
Alloimmune hepatitis;
Alopecia areata;
Alpers disease;
Alveolar proteinosis;
Ammonia abnormal;
Ammonia increased;
Amniotic cavity infection;
Amygdalohippocampectomy;
Amyloid arthropathy;
Amyloidosis;
Amyloidosis senile;
Anaphylactic reaction;
Anaphylactic shock;
Anaphylactic transfusion reaction;
Anaphylactoid reaction;
Anaphylactoid shock;
________________________________________________________________________
Anaphylactoid syndrome of pregnancy;
________________________________________________________________________
Angioedema;
Angiopathic neuropathy;
Ankylosing spondylitis;
Anosmia;
Antiacetylcholine receptor antibody positive;
Anti-actin antibody positive;
Anti-aquaporin-4 antibody positive;
Anti-basal ganglia antibody positive;
Anti-cyclic citrullinated peptide antibody positive;
Anti-epithelial antibody positive;
Anti-erythrocyte antibody positive;
Anti-exosome complex antibody positive;
Anti-GAD antibody negative;
Anti-GAD antibody positive;
Anti-ganglioside antibody
positive;
Antigliadin antibody positive;
Anti-glomerular basement membrane antibody
positive;
Anti-glomerular basement membrane disease;
Anti-glycyl-tRNA synthetase antibody positive;
Anti-HLA antibody test positive;
Anti-IA2 antibody positive;
Anti-insulin antibody increased;
Anti-insulin antibody positive;
Anti-insulin receptor antibody increased;
Anti-insulin receptor antibody positive;
Anti-interferon antibody negative;
Anti-interferon antibody positive;
Anti-islet cell antibody positive;
Antimitochondrial antibody positive;
Anti-muscle specific kinase antibody positive;
Anti-myelin-associated glycoprotein antibodies positive;
Anti-myelin-associated glycoprotein associated polyneuropathy;
Antimyocardial antibody positive;
Anti-neuronal antibody positive;
Antineutrophil cytoplasmic antibody increased;
Antineutrophil cytoplasmic antibody positive;
Anti-neutrophil cytoplasmic antibody positive vasculitis;
Anti-NMDA antibody positive;
Antinuclear antibody increased;
Antinuclear antibody positive;
Antiphospholipid antibodies
positive;Antiphospholipid syndrome;
Anti-platelet antibody positive;
Anti-prothrombin antibody positive;
Antiribosomal P antibody positive;
Anti-RNA polymerase III antibody positive;
Anti-saccharomyces cerevisiae antibody test positive;
Anti-sperm antibody positive;
Anti-SRP antibody positive;
Antisynthetase syndrome;
Anti-thyroid antibody positive;
Anti-transglutaminase antibody increased;
Anti-VGCC antibody positive;
Anti-VGKC antibody positive;
Anti-vimentin antibody positive;
Antiviral prophylaxis;
Antiviral treatment;
Anti-zinc transporter 8 antibody positive;
Aortic embolus;Aortic thrombosis;
Aortitis;
Aplasia pure red cell;
Aplastic anaemia;
Application site thrombosis;
Application site vasculitis;
Arrhythmia;Arterial bypass occlusion;
Arterial bypass thrombosis;
Arterial thrombosis;
Arteriovenous fistula thrombosis;
Arteriovenous graft site stenosis;Arteriovenous graft thrombosis;
Arteritis;Arteritis
________________________________________________________________________
Page 2;
________________________________________________________________________
coronary;
Arthralgia;
Arthritis;
Arthritis enteropathic;
Ascites;
Aseptic cavernous sinus thrombosis;
Aspartate aminotransferase abnormal;
Aspartate aminotransferase increased;
Aspartate-glutamate-transporter deficiency;
AST to platelet ratio index increased;
AST/ALT ratio abnormal;
Asthma;
Asymptomatic COVID-19;
Ataxia;
Atheroembolism;
Atonic seizures;
Atrial thrombosis;
Atrophic thyroiditis;
Atypical benign partial epilepsy;
Atypical pneumonia;
Aura;
Autoantibody positive;
Autoimmune anaemia;
Autoimmune aplastic anaemia;
Autoimmune arthritis;
Autoimmune blistering disease;
Autoimmune cholangitis;
Autoimmune colitis;
Autoimmune demyelinating disease;
Autoimmune dermatitis;
Autoimmune disorder;
Autoimmune encephalopathy;
Autoimmune endocrine disorder;
Autoimmune enteropathy;
Autoimmune eye disorder;
Autoimmune haemolytic anaemia;
Autoimmune heparin-induced thrombocytopenia;
Autoimmune hepatitis;
Autoimmune hyperlipidaemia;
Autoimmune hypothyroidism;
Autoimmune inner ear disease;
Autoimmune lung disease;
Autoimmune lymphoproliferative syndrome;
Autoimmune myocarditis;
Autoimmune myositis;
Autoimmune nephritis;
Autoimmune neuropathy;
Autoimmune neutropenia;
Autoimmune pancreatitis;
Autoimmune pancytopenia;
Autoimmune pericarditis;
Autoimmune retinopathy;
Autoimmune thyroid disorder;
Autoimmune thyroiditis;
Autoimmune uveitis;
Autoinflammation with infantile enterocolitis;
Autoinflammatory disease;
Automatism epileptic;
Autonomic nervous system imbalance;
Autonomic seizure;
Axial spondyloarthritis;
Axillary vein thrombosis;
Axonal and demyelinating polyneuropathy;
Axonal neuropathy;
Bacterascites;
Baltic myoclonic epilepsy;
Band sensation;
Basedow's disease;
Basilar artery thrombosis;
Basophilopenia;
B-cell aplasia;
Behcet's syndrome;
Benign ethnic neutropenia;
Benign familial neonatal convulsions;
Benign familial pemphigus;
Benign rolandic epilepsy;
Beta-2 glycoprotein antibody positive;
Bickerstaff's encephalitis;
Bile output abnormal;
Bile output decreased;
Biliary ascites;
Bilirubin conjugated abnormal;
Bilirubin conjugated increased;
Bilirubin urine present;
Biopsy liver abnormal;
Biotinidase deficiency;
Birdshot chorioretinopathy;
Blood alkaline phosphatase abnormal;
Blood alkaline phosphatase increased;
Blood bilirubin abnormal;
Blood bilirubin increased;
Blood bilirubin unconjugated increased;
Blood cholinesterase abnormal;
Blood cholinesterase decreased;
Blood pressure decreased;
Blood pressure diastolic decreased;
Blood pressure systolic decreased;
Blue toe syndrome;
Brachiocephalic vein thrombosis;
Brain stem embolism;
Brain stemn thrombosis;
Bromosulphthalein test abnormal;
Bronchial oedema;
Bronchitis;
Bronchitis mycoplasmal;
Bronchitis viral;
Bronchopulmonary aspergillosis allergic;
Bronchospasm;Budd-Chiari syndrome;
Bulbar palsy;
Butterfly rash;
C1q nephropathy;
Caesarean section;
Calcium embolism;
Capillaritis;
Caplan's syndrome;
Cardiac amyloidosis;
Cardiac arrest;
Cardiac failure;
Cardiac failure acute;
Cardiac sarcoidosis;
Cardiac ventricular thrombosis;
Cardiogenic shock;
Cardiolipin antibody positive;
Cardiopulmonary failure;
Cardio-respiratory arrest;
Cardio-respiratory distress;
Cardiovascular insufficiency;
Carotid arterial embolus;
Carotid artery thrombosis;
Cataplexy;
Catheter site thrombosis;
Catheter site vasculitis;
Cavernous sinus thrombosis;
CDKL5 deficiency disorder;
CEC syndrome;
Cementembolism;
Central nervous system lupus;
Central nervous system vasculitis;
Cerebellar artery thrombosis;
Cerebellar embolism;
Cerebral amyloid angiopathy;
Cerebral arteritis;
Cerebral artery embolism;
Cerebral artery thrombosis;
Cerebral gas embolism;
Cerebral microembolism;
Cerebral septic infarct;
Cerebral thrombosis;
Cerebral venous sinus thrombosis;
Cerebral venous thrombosis;
Cerebrospinal thrombotic
________________________________________________________________________
Page 3;
________________________________________________________________________
tamponade;
Cerebrovascular accident;
Change in seizure presentation;
Chest discomfort;
________________________________________________________________________
Child-Pugh-Turcotte score abnormal;
Child-Pugh-Turcotte score increased;
________________________________________________________________________
Chillblains;
Choking;Choking sensation;
Cholangitis sclerosing;
Chronic autoimmune glomerulonephritis;
Chronic cutaneous lupus erythematosus;
Chronic fatigue syndrome;
Chronic gastritis;
Chronic inflammatory demyelinating polyradiculoneuropathy;
Chronic lymphocytic inflammation with pontine perivascular enhancement responsive to steroids;
Chronic recurrent multifocal osteomyelitis;
Chronic respiratory failure;
Chronic spontaneous urticaria;
Circulatory collapse;
Circumoral oedema;
Circumoral swelling;
Clinically isolated syndrome;
Clonic convulsion;
Coeliac disease;
Cogan's syndrome;
Cold agglutinins positive;
Cold type haemolytic anaemia;
Colitis;
Colitis erosive;
Colitis herpes;
Colitis microscopic;
Colitis ulcerative;
Collagen disorder;
Collagen-vascular disease;
Complement factor abnormal;
Complement factor C1 decreased;
Complement factor C2 decreased;
Complement factor C3 decreased;
Complement factor C4 decreased;
Complement factor decreased;
Computerised tomogram liver abnormal;
Concentric sclerosis;Congenital anomaly;
Congenital bilateral perisylvian syndrome;
Congenital herpes simplex infection;
Congenital myasthenic syndrome;
Congenital varicella infection;
Congestive hepatopathy;
Convulsion in childhood;
Convulsions local;
Convulsive threshold lowered;
Coombs positive haemolytic anaemia;
Coronary artery disease;
Coronary artery embolism;
Coronary artery thrombosis;
Coronary bypass thrombosis;
Coronavirus infection;
Coronavirus test;
Coronavirus test negative;
Coronavirus test positive;
Corpus callosotomy;
Cough;Cough variant asthma;
COVID-19;
COVID-19 immunisation;
COVID-19 pneumonia;
COVID-19 prophylaxis;
COVID-19 treatment;
Cranial nerve disorder;
Cranial nerve palsies multiple;
Cranial nerve paralysis;
CREST syndrome;
Crohn's disease;
Cryofibrinogenaemia;
Cryoglobulinaemia;
CSF oligoclonal band present;
CSWS syndrome;Cutaneous amyloidosis;
Cutaneous lupus erythematosus;
Cutaneous sarcoidosis;
Cutaneous vasculitis;
Cyanosis;
Cyclic neutropenia;
Cystitis interstitial;
Cytokine release syndrome;
Cytokine storm;
De novo purine synthesis inhibitors associated acute inflammatory syndrome;
Death neonatal;
Deep vein thrombosis;
Deep vein thrombosis postoperative;
Deficiency of bile secretion;
Deja vu;
Demyelinating polyneuropathy;
Demyelination;
Dermatitis;
Dermatitis bullous;
Dermatitis herpetiformis;
Dermatomyositis;Device embolisation;
Device related thrombosis;
Diabetes mellitus;
Diabetic ketoacidosis;
Diabetic mastopathy;
Dialysis amyloidosis;
Dialysis membrane reaction;
Diastolic hypotension;
Diffuse vasculitis;
Digital pitting scar;
Disseminated intravascular coagulation;
Disseminated intravascular coagulation in newborn;
Disseminated neonatal herpes simplex;
Disseminated varicella;
Disseminated varicella zoster vaccine virus infection;
Disseminated varicella zoster virus infection;
DNA antibody positive;
Double cortex syndrome;
Double stranded DNA antibody positive;
Dreamy state;
Dressler's syndrome;
Drop attacks;
Drug withdrawal convulsions;
Dyspnoea;Early infantile epileptic encephalopathy with burst-suppression;
Eclampsia;Eczema herpeticum;Embolia cutis medicamentosa;
Embolic cerebellar infarction;
Embolic cerebral infarction;
Embolic pneumonia;
Embolic stroke;
Embolism;
Embolism arterial;
Embolism venous;
Encephalitis;
Encephalitis allergic;
Encephalitis autoimmune;
Encephalitis brain stem;
Encephalitis haemorrhagic;
Encephalitis periaxialis diffusa;
Encephalitis post immunisation;
Encephalomyelitis;
Encephalopathy;
Endocrine disorder;
Endocrine ophthalmopathy;
Endotracheal intubation;
Enteritis;
Enteritis leukopenic;
Enterobacter pneumonia;
Enterocolitis;
Enteropathic
________________________________________________________________________
Page 4;
________________________________________________________________________
fasciitis;
Eosinophilic granulomatosis with polyangiitis;
Eosinophilic oesophagitis;
Epidermolysis;
Epilepsy;
Epilepsy surgery;
Epilepsy with myoclonic-atonic seizures;
Epileptic aura;
Epileptic psychosis;
Erythema;Erythema induratum;
Erythema multiforme;
Erythema nodosum;
Evans syndrome;
Exanthema subitum;
Expanded disability status scale score decreased;
Expanded disability status scale score increased;
Exposure to communicable disease;
Exposure to SARS-CoV-2;
Eye oedema;
Eye pruritus;
Eye swelling;
Eyelid oedema;
Face oedema;
Facial paralysis;
Facial paresis;
Faciobrachial dystonic seizure;
Fat embolism;
Febrile convulsion;
Febrile infection-related epilepsy syndrome;
Febrile neutropenia;
Felty's syndrome;
Femoral artery embolism;
Fibrillary glomerulonephritis;
Fibromyalgia;
Flushing;
Foaming at mouth;
Focal cortical resection;
Focal dyscognitive seizures;
Foetal distress syndrome;
Foetal placental thrombosis;
Foetor hepaticus;
Foreign body embolism;
Frontal lobe epilepsy;
Fulminant type 1 diabetes mellitus;
Galactose elimination capacity test abnormal;
Galactose elimination capacity test decreased;
Gamma-glutamyltransferase abnormal;
Gamma-glutamyltransferase increased;
Gastritis herpes;
Gastrointestinal amyloidosis;
Gelastic seizure;
Generalised onset non-motor seizure;
Generalised tonic-clonic seizure;
Genital herpes;
Genital herpes simplex;
Genital herpes zoster;
Giant cell arteritis;
Glomerulonephritis;
Glomerulonephritis membranoproliferative;
Glomerulonephritis membranous;
Glomerulonephritis rapidly progressive;
Glossopharyngeal nerve paralysis;
Glucose transporter type 1 deficiency syndrome;
Glutamate dehydrogenase increased;
Glycocholic acid increased;
GM2 gangliosidosis;
Goodpasture's syndrome;
Graft thrombosis;
Granulocytopenia;
Granulocytopenia neonatal;
Granulomatosis with polyangiitis;
Granulomatous dermatitis;
Grey matter heterotopia;
Guanase increased;
Guillain-Barre syndrome;
Haemolytic anaemia;
Haemophagocytic lymphohistiocytosis;
Haemorrhage;
Haemorrhagic ascites;
Haemorrhagic disorder;
Haemorrhagic pneumonia;
Haemorrhagic varicella syndrome;
Haemorrhagic vasculitis;
Hantavirus pulmonary infection;
Hashimoto's encephalopathy;
Hashitoxicosis;
Hemimegalencephaly;
Henoch-Schonlein purpura;
Henoch-Schonlein purpura nephritis;
Hepaplastin abnormal;
Hepaplastin decreased;
Heparin-induced thrombocytopenia;
Hepatic amyloidosis;
Hepatic artery embolism;
Hepatic artery flow decreased;
Hepatic artery thrombosis;
Hepatic enzyme abnormal;
Hepatic enzyme decreased;
Hepatic enzyme increased;
Hepatic fibrosis marker abnormal;
Hepatic fibrosis marker increased;
Hepatic function abnormal;
Hepatic hydrothorax;
Hepatic hypertrophy;
Hepatic hypoperfusion;
Hepatic lymphocytic infiltration;
Hepatic mass;
Hepatic pain;
Hepatic sequestration;
Hepatic vascular resistance increased;
Hepatic vascular thrombosis;
Hepatic vein embolism;
Hepatic vein thrombosis;
Hepatic venous pressure gradient abnormal;
Hepatic venous pressure gradient increased;
Hepatitis;
Hepatobiliary scan abnormal;
Hepatomegaly;
Hepatosplenomegaly;
Hereditary angioedema with C1 esterase inhibitor deficiency;
Herpes dermatitis;
Herpes gestationis;
Herpes oesophagitis;
Herpes ophthalmic;
Herpes pharyngitis;
Herpes sepsis;
Herpes simplex;
Herpes simplex
cervicitis;
Herpes simplex colitis;
Herpes simplex encephalitis;
Herpes simplex gastritis;
Herpes simplex hepatitis;
Herpes simplex meningitis;
Herpes simplex meningoencephalitis;
Herpes simplex meningomyelitis;
Herpes simplex necrotising retinopathy;
Herpes simplex oesophagitis;
Herpes simplex otitis externa;
Herpes simplex pharyngitis;
Herpes simplex pneumonia;
Herpes simplex reactivation;
Herpes simplex sepsis;
Herpes simplex viraemia;
Herpes simplex virus conjunctivitis neonatal;
Herpes simplex visceral;
Herpes virus
________________________________________________________________________
Page 5;
________________________________________________________________________
infection;
Herpes zoster;
Herpes zoster cutaneous disseminated;
Herpes zoster infection neurological;
Herpes zoster meningitis;
Herpes zoster meningoencephalitis;
Herpes zoster meningomyelitis;
Herpes zoster meningoradiculitis;
Herpes zoster necrotising retinopathy;
Herpes zoster oticus;
Herpes zoster pharyngitis;
Herpes zoster reactivation;
Herpetic radiculopathy;
Histone antibody positive;
Hoigne's syndrome;
Human herpesvirus 6 encephalitis;
Human herpesvirus 6 infection;
Human herpesvirus 6 infection reactivation;
Human herpesvirus 7 infection;
Human herpesvirus 8 infection;
Hyperammonaemia;
Hyperbilirubinaemia;
Hypercholia;
Hypergammaglobulinaemia benign monoclonal;
Hyperglycaemic seizure;
Hypersensitivity;
Hypersensitivity vasculitis;
Hyperthyroidism;
Hypertransaminasaemia;
Hyperventilation;
Hypoalbuminaemia;
Hypocalcaemic seizure;
Hypogammaglobulinaemia;
Hypoglossal nerve paralysis;
Hypoglossal nerve paresis;
Hypoglycaemic seizure;
Hyponatraemic seizure;
Hypotension;
Hypotensive crisis;
Hypothenar hammer syndrome;
Hypothyroidism;
Hypoxia;
Idiopathic CD4 lymphocytopenia;
Idiopathic generalised epilepsy;
Idiopathic interstitial pneumonia;
Idiopathic neutropenia;
Idiopathic pulmonary fibrosis;
IgA nephropathy;
IgM nephropathy;
IIIrd nerve paralysis;
IIIrd nerve paresis;
Iliac artery embolism;
Immune thrombocytopenia;
Immune-mediated adverse reaction;
Immune-mediated cholangitis;
Immune-mediated cholestasis;
Immune-mediated cytopenia;
Immune-mediated encephalitis;
Immune-mediated encephalopathy;
Immune-mediated endocrinopathy;
Immune-mediated enterocolitis;
Immune-mediated gastritis;
Immune-mediated hepatic disorder;
Immune-mediated hepatitis;
Immune-mediated hyperthyroidism;
Immune-mediated hypothyroidism;
Immune-mediated myocarditis;
Immune-mediated myositis;
Immune-mediated nephritis;
Immune-mediated neuropathy;
Immune-mediated pancreatitis;
Immune-mediated pneumonitis;
Immune-mediated renal disorder;
Immune-mediated thyroiditis;
Immune-mediated uveitis;
Immunoglobulin G4 related disease;
Immunoglobulins abnormal;
Implant site thrombosis;
Inclusion body myositis;
Infantile genetic agranulocytosis;
Infantile spasms;
Infected vasculitis;
Infective thrombosis;
Inflammation;
Inflammatory bowel disease;
Infusion site thrombosis;
Infusion site
vasculitis;
Injection site thrombosis;
Injection site urticaria;
Injection site vasculitis;
Instillation site thrombosis;
Insulin autoimmune syndrome;
Interstitial granulomatous dermatitis;
Interstitial lung disease;Intracardiac mass;
Intracardiac thrombus;
Intracranialn pressure increased;
Intrapericardial thrombosis;
Intrinsic factor antibody abnormal;
Intrinsic factor antibody positive;
IPEX syndrome;
Irregular breathing;
IRVAN syndrome;
IVth nerve paralysis;
IVth nerve paresis;
JC polyomavirus test positive;
JC virus CSF test positive;
Jeavons syndrome;
Jugular vein embolism;
Jugular vein thrombosis;
Juvenile idiopathic arthritis;
Juvenile myoclonic epilepsy;
Juvenile polymyositis;
Juvenile psoriatic arthritis;
Juvenile spondyloarthritis;
Kaposi sarcoma inflammatory cytokine syndrome;
Kawasaki's disease;
Kayser-Fleischer ring;
Keratoderma blenorrhagica;
Ketosis-prone diabetes mellitus;
Kounis syndrome;
Lafora's myoclonic epilepsy;
Lambl's excrescences;
Laryngeal dyspnoea;
Laryngeal oedema;
Laryngeal rheumatoid arthritis;
Laryngospasm;
Laryngotracheal oedema;
Latent autoimmune diabetes in adults;
LEcells present;
Lemierre syndrome;
Lennox-Gastaut syndrome;
Leucine aminopeptidase increased;
Leukoencephalomyelitis;
Leukoencephalopathy;
Leukopenia;
Leukopenia neonatal;
Lewis-Sumner syndrome;
Lhermitte's sign;
Lichen planopilaris;
Lichen planus;
Lichen sclerosus;
Limbic encephalitis;
Linear IgA disease;
Lip oedema;
Lip swelling;
Liver function test abnormal;
Liver function test decreased;
Liver function test increased;
Liver induration;
Liver injury;
Liver iron concentration abnormal;
Liver iron concentration
________________________________________________________________________
Page 6:
________________________________________________________________________
increased;
Liver opacity;
Liver palpable;
Liver sarcoidosis;
Liver scan abnormal;
Liver tenderness;
________________________________________________________________________
Low birth weight baby;
________________________________________________________________________
Lower respiratory tract herpes infection;
Lower respiratory tract infection;
Lower respiratory tract infection viral;
Lung abscess;
Lupoid hepatic cirrhosis;
Lupus cystitis;
Lupus encephalitis;
Lupus endocarditis;
Lupus enteritis;
Lupus hepatitis;
Lupus myocarditis;
Lupus myositis;
Lupus nephritis;
Lupus pancreatitis;
Lupus pleurisy;
Lupus pneumonitis;
Lupus vasculitis;
Lupus-like syndrome;
Lymphocytic hypophysitis;
Lymphocytopenia neonatal;
Lymphopenia;
MAGIC syndrome;
Magnetic resonance imaging liver abnormal;
Magnetic resonance proton density fat fraction measurement;
Mahler sign;
Manufacturing laboratory analytical testing issue;
Manufacturing materials issue;
Manufacturing production issue;
Marburg's variant multiple sclerosis;
Marchiafava-Bignami disease;
Marine Lenhart syndrome;
Mastocytic enterocolitis;
________________________________________________________________________
Maternal exposure during pregnancy;
________________________________________________________________________
Medical device site thrombosis;
Medical device site vasculitis;
MELAS syndrome;
Meningitis;
Meningitis aseptic;
Meningitis herpes;
Meningoencephalitis herpes simplex neonatal;
Meningoencephalitis herpetic;
Meningomyelitis herpes;
MERS-CoV test;
MERS-CoV test negative;
MERS-CoV test positive;
Mesangioproliferative glomerulonephritis;
Mesenteric artery embolism;
Mesenteric artery thrombosis;
Mesenteric vein thrombosis;
Metapneumovirus infection;
Metastatic cutaneous Crohn's disease;
Metastatic pulmonary embolism;
Microangiopathy;
Microembolism;
Microscopic polyangiitis;
Middle East respiratory syndrome;
Migraine-triggered seizure;
Miliary pneumonia;
Miller Fisher syndrome;
Mitochondrial aspartate aminotransferase increased;
Mixed connective tissue disease;
Model for end stage liver disease score abnormal;
Model for end stage liver disease score increased;
Molar ratio of total branched-chain amino acid to tyrosine;
Molybdenum cofactor deficiency;
Monocytopenia;
Mononeuritis;
Mononeuropathy multiplex;
Morphoea;
Morvan syndrome;
Mouth swelling;
Moyamoya disease;
Multifocal motor neuropathy;
Multiple organ dysfunction syndrome;
Multiple sclerosis;
Multiple sclerosis relapse;
Multiple sclerosis relapse prophylaxis;
Multiple subpial transection;
________________________________________________________________________
Multisystem inflammatory syndrome in children;
________________________________________________________________________
Muscular sarcoidosis;
Myasthenia gravis;
Myasthenia gravis crisis;
Myasthenia gravis neonatal;
Myasthenic syndrome;
Myelitis;
Myelitis transverse;
Myocardial infarction;
Myocarditis;Myocarditis post infection;
Myoclonic epilepsy;
Myoclonic epilepsy and ragged-red fibres;
Myokymia;
Myositis;
Narcolepsy;
Nasal herpes;
Nasal obstruction;
Necrotising herpetic retinopathy;
Neonatal Crohn's disease;
Neonatal epileptic seizure;
Neonatal lupus erythematosus;
Neonatal mucocutaneous herpes simplex;
Neonatal pneumonia;Neonatal seizure;
Nephritis;
Nephrogenic systemic fibrosis;
Neuralgic amyotrophy;
Neuritis;
Neuritis cranial;
Neuromyelitis optica pseudo relapse;
Neuromyelitis optica spectrum disorder;
Neuromyotonia;
Neuronal neuropathy;
Neuropathy peripheral;
Neuropathy, ataxia, retinitis pigmentosa syndrome;
Neuropsychiatric lupus;
Neurosarcoidosis;
Neutropenia;
Neutropenia neonatal;
Neutropenic colitis;
Neutropenic infection;
Neutropenic sepsis;
Nodular rash;
Nodular vasculitis;
Noninfectious myelitis;
Noninfective encephalitis;
Noninfective encephalomyelitis;
Noninfective oophoritis;
Obstetrical pulmonary embolism;
Occupational exposure to communicable disease;
Occupational exposure to SARS-CoV-2;
Ocular hyperaemia;
Ocular myasthenia;
Ocular pemphigoid;
Ocular sarcoidosis;
Ocular vasculitis;
Oculofacial paralysis;
Oedema;
Oedema blister;
Oedema due to hepatic disease;
Oedema mouth;
Oesophageal achalasia;
Ophthalmic artery thrombosis;
Ophthalmic herpes simplex;
Ophthalmic herpes zoster;
Ophthalmic vein thrombosis;
Optic neuritis;
Optic
________________________________________________________________________
Page 7;
________________________________________________________________________
neuropathy;
Optic perineuritis;
Oral herpes;Oral lichen planus;
Oropharyngeal oedema;
Oropharyngeal spasm;
Oropharyngeal swelling;
Osmotic demyelination syndrome;
Ovarian vein thrombosis;
Overlap syndrome;
Paediatric autoimmune neuropsychiatric disorders associated with streptococcal infection;
Paget-Schroetter syndrome;
Palindromic rheumatism;
Palisaded neutrophilic granulomatous dermatitis;
Palmoplantar keratoderma;
Palpable purpura;
Pancreatitis;
Panencephalitis;
Papillophlebitis;
Paracancerous pneumonia;
Paradoxical embolism;
Parainfluenzae viral laryngotracheobronchitis;
Paraneoplastic dermatomyositis;
Paraneoplastic pemphigus;
Paraneoplastic thrombosis;
Paresis cranial nerve;
Parietal cell antibody positive;
Paroxysmal nocturnal haemoglobinuria;
Partial seizures;
Partial seizures with secondary generalisation;
Patient isolation;
Pelvic venous thrombosis;
Pemphigoid;
Pemphigus;
Penile vein thrombosis;
Pericarditis;
Pericarditis lupus;
Perihepatic discomfort;
Periorbital oedema;
Periorbital swelling;
Peripheral artery thrombosis;
Peripheral embolism;
Peripheral ischaemia;
Peripheral vein thrombus extension;
Periportal oedema;
Peritoneal fluid protein abnormal;
Peritoneal fluid protein decreased;
Peritoneal fluid protein increased;
Peritonitis lupus;
Pernicious anaemia;
Petit mal epilepsy;
Pharyngeal oedema;
Pharyngeal swelling;
Pityriasis lichenoides et varioliformis acuta;
Placenta praevia;
Pleuroparenchymal fibroelastosis;
Pneumobilia;Pneumonia;
Pneumonia adenoviral;
Pneumonia cytomegaloviral;
Pneumonia herpes viral;
Pneumonia influenzal;
Pneumonia measles;
Pneumonia mycoplasmal;
Pneumonia necrotising;
Pneumonia parainfluenzae viral;
Pneumonia respiratory syncytial viral;
Pneumonia viral;
POEMS syndrome;
Polyarteritis nodosa;
Polyarthritis;
Polychondritis;
Polyglandular autoimmune syndrome type I;
Polyglandular autoimmune syndrome type II;
Polyglandular autoimmune syndrome type III;
Polyglandular disorder;
Polymicrogyria;
Polymyalgia rheumatica;
Polymyositis;
Polyneuropathy;
Polyneuropathy idiopathic progressive;
Portal pyaemia;
Portal vein embolism;
Portal vein flow decreased;
Portal vein pressure increased;
Portal vein thrombosis;
Portosplenomesenteric venous thrombosis;
Post procedural hypotension;
Post procedural pneumonia;
Post procedural pulmonary embolism;
Post stroke epilepsy;
Post stroke seizure;
Post thrombotic retinopathy;
Post thrombotic syndrome;
Post viral fatigue syndrome;
Postictal headache;Postictal paralysis;
Postictal psychosis;
Postictal state;
Postoperative respiratory distress;
Postoperative respiratory failure;
Postoperative thrombosis;
Postpartum thrombosis;
Postpartum venous thrombosis;
Postpericardiotomy syndrome;
Post-traumatic epilepsy;
Postural orthostatic tachycardia syndrome;
Precerebral artery thrombosis;
Pre-eclampsia;Preictal state;
Premature labour;
Premature menopause;
Primary amyloidosis;
Primary biliary cholangitis;
Primary progressive multiple
sclerosis;
Procedural shock;
Proctitis herpes;
Proctitis ulcerative;
Product availability issue;
Product distribution issue;
Product supply issue;
Progressive facial
hemiatrophy;
Progressive multifocal leukoencephalopathy;
Progressive multiple sclerosis;
Progressive relapsing multiple sclerosis;
Prosthetic cardiac valve thrombosis;
Pruritus;
Pruritus allergic;
Pseudovasculitis;
Psoriasis;Psoriatic
arthropathy;
Pulmonary amyloidosis;
Pulmonary artery thrombosis;
Pulmonary embolism;
Pulmonary fibrosis;
Pulmonary haemorrhage;
Pulmonary microemboli;
Pulmonary oil microembolism;
Pulmonary renal syndrome;
Pulmonary sarcoidosis;
Pulmonary sepsis;
Pulmonary thrombosis;
Pulmonary tumour thrombotic microangiopathy;
Pulmonary vasculitis;
Pulmonary veno-occlusive disease;
Pulmonary venous thrombosis;
Pyoderma gangrenosum;
Pyostomatitis vegetans;
Pyrexia;
Quarantine;
Radiation leukopenia;
Radiculitis
________________________________________________________________________
Page 8;
________________________________________________________________________
brachial;
Radiologically isolated syndrome;
Rash;
Rash erythematous;
Rash pruritic;
Rasmussen
encephalitis;
Raynaud's phenomenon;
Reactive capillary endothelial proliferation;
Relapsing multiple sclerosis;
Relapsing-remitting multiple sclerosis;
Renal amyloidosis;
Renal arteritis;
Renal artery thrombosis;
Renal embolism;Renal failure;
Renal vascular thrombosis;
Renal vasculitis;
Renal vein embolism;
Renal vein thrombosis;
Respiratory arrest;
Respiratory disorder;
Respiratory distress;
Respiratory failure;
Respiratory paralysis;
Respiratory syncytial virus bronchiolitis;
Respiratory syncytial virus bronchitis;
Retinal artery embolism;
Retinal artery occlusion;
Retinal artery thrombosis;
Retinal vascular thrombosis;
Retinal vasculitis;
Retinal vein occlusion;
Retinal vein thrombosis;
Retinol binding protein decreased;
Retinopathy;
Retrograde portal vein flow;
Retroperitoneal fibrosis;
Reversible airways obstruction;
Reynold's syndrome;
Rheumatic brain disease;
Rheumatic disorder;
Rheumatoid arthritis;
Rheumatoid factor increased;
Rheumatoid factor positive;
Rheumatoid factor quantitative increased;
Rheumatoid lung;
Rheumatoid neutrophilic dermatosis;
Rheumatoid nodule;
Rheumatoid nodule removal;
Rheumatoid scleritis;
Rheumatoid vasculitis;
Saccadic eye movement;
SAPHO syndrome;
Sarcoidosis;
SARS-CoV-1 test;
SARS-CoV-1 test negative;
SARS-CoV-1 test positive;
SARS-CoV-2 antibody test;
SARS-CoV-2 antibody test negative;
SARS-CoV-2 antibody test positive;
SARS-CoV-2 carrier;
SARS-CoV-2 sepsis;
SARS-CoV-2 test;
SARS- CoV-2 test false negative;
SARS-CoV-2 test false positive;
SARS-CoV-2 test negative;
SARS-CoV-2 test positive;
SARS-CoV-2 viraemia;
Satoyoshi syndrome;
Schizencephaly;
Scleritis;
Sclerodactylia;
Scleroderma;
Scleroderma associated digital ulcer;
Scleroderma renal crisis;
Scleroderma-like reaction;
Secondary amyloidosis;
Secondary cerebellar degeneration;
Secondary progressive multiple sclerosis;
Segmented hyalinising vasculitis;
Seizure;
Seizure anoxic;
Seizure cluster;
Seizure like phenomena;
Seizure prophylaxis;
Sensation of foreign body;
Septic embolus;
Septic pulmonary embolism;
Severe acute respiratory syndrome;
________________________________________________________________________
Severe myoclonic epilepsy of infancy;
Shock;
Shock symptom;
Shrinking lung syndrome;
Shunt thrombosis;
Silent thyroiditis;
Simple partial seizures;
Sjogren's syndrome;
Skin swelling;
SLE arthritis;
Smooth muscle antibody positive;
Sneezing;Spinal artery embolism;
Spinal artery thrombosis;
Splenic artery thrombosis;
Splenic embolism;
Splenic thrombosis;
Splenic vein thrombosis;
Spondylitis;
Spondyloarthropathy;
Spontaneous heparin-induced thrombocytopenia syndrome;
Status epilepticus;
Stevens-Johnson syndrome;
Stiff leg syndrome;
Stiff person syndrome;
Stillbirth;
Still's disease;
Stoma site thrombosis;
Stoma site vasculitis;
Stress cardiomyopathy;
Stridor;
Subacute cutaneous lupus erythematosus;
Subacute endocarditis;
Subacute inflammatory demyelinating polyneuropathy;
Subclavian artery embolism;
Subclavian artery thrombosis;
Subclavian vein thrombosis;
Sudden unexplained death in epilepsy;
Superior sagittal sinus thrombosis;
Susac's syndrome;Suspected COVID-19;
Swelling;
Swelling face;
Swelling of eyelid;
Swollen tongue;
Sympathetic ophthalmia;
Systemic lupus erythematosus;
Systemic lupus erythematosus disease activity index abnormal;
Systemic lupus erythematosus disease activity index decreased;
Systemic lupus erythematosus disease activity index increased;
Systemic lupus erythematosus rash;
Systemic scleroderma;
Systemic sclerosis pulmonary;
Tachycardia;
Tachypnoea;
Takayasu's arteritis;
Temporal lobe epilepsy;
Terminal ileitis;
Testicular autoimmunity;
Throat tightness;
Thromboangiitis obliterans;
Thrombocytopenia;
Thrombocytopenic purpura;
Thrombophlebitis;
Thrombophlebitis migrans;
Thrombophlebitis
________________________________________________________________________
Page 9;
________________________________________________________________________
neonatal;
Thrombophlebitis septic;
Thrombophlebitis superficial;
Thromboplastin antibody positive;
Thrombosis;Thrombosis corpora cavernosa;
Thrombosis in device;
Thrombosis mesenteric vessel;
Thrombotic cerebral infarction;
Thrombotic microangiopathy;
Thrombotic stroke;
Thrombotic thrombocytopenic purpura;
Thyroid disorder;
Thyroid stimulating immunoglobulin increased;
Thyroiditis;
Tongue amyloidosis;
Tongue biting;
Tongue oedema;
Tonic clonic movements;
Tonic convulsion;
Tonic posturing;
Topectomy;
Total bile acids increased;
Toxic epidermal necrolysis;
Toxic leukoencephalopathy;
Toxic oil syndrome;
Tracheal obstruction;
Tracheal oedema;
Tracheobronchitis;
Tracheobronchitis mycoplasmal;
Tracheobronchitis viral;
Transaminases abnormal;
Transaminases increased;
Transfusion-related alloimmune neutropenia;
Transient epileptic amnesia;Transverse sinus thrombosis;
Trigeminal nerve paresis;
Trigeminal neuralgia;
Trigeminal palsy;Truncus coeliacus thrombosis;
Tuberous sclerosis complex;
Tubulointerstitial nephritis and uveitis syndrome;
Tumefactive multiple sclerosis;
Tumour embolism;
Tumour thrombosis;
Type 1 diabetes mellitus;
Type I hypersensitivity;
Type III immune complex mediated reaction;
Uhthoff's phenomenon;Ulcerative keratitis;Ultrasound liver abnormal;
________________________________________________________________________
Umbilical cord thrombosis;
________________________________________________________________________
Uncinate fits;
Undifferentiated connective tissue disease;
Upper airway obstruction;
Urine bilirubin increased;
Urobilinogen urine decreased;
Urobilinogen urine increased;
Urticaria;
Urticaria papular;
Urticarial vasculitis;
Uterine rupture;
Uveitis;
Vaccination site thrombosis;
Vaccination site vasculitis;
Vagus nerve paralysis;
Varicella;
Varicella keratitis;
Varicella post vaccine;
Varicella zoster gastritis;
Varicella zoster oesophagitis;
Varicella zoster pneumonia;
Varicella zoster sepsis;
Varicella zoster virus infection;
Vasa praevia;Vascular graft thrombosis;
Vascular pseudoaneurysm thrombosis;
Vascular purpura;
Vascular stent thrombosis;
Vasculitic rash;
Vasculitic ulcer;
Vasculitis;
Vasculitis gastrointestinal;
Vasculitis necrotising;
Vena cava embolism;
Vena cava thrombosis;
Venous intravasation;
Venous recanalisation;
________________________________________________________________________
Venous thrombosis;
Venous thrombosis in pregnancy;
________________________________________________________________________
Venous thrombosis limb;
Venous thrombosis neonatal;
Vertebral artery thrombosis;
Vessel puncture site thrombosis;
Visceral venous thrombosis;
VIth nerve paralysis;
VIth nerve paresis;
Vitiligo;
Vocal cord paralysis;
Vocal cord paresis;
Vogt-Koyanagi-Harada disease;
Warm type haemolytic anaemia;
Wheezing;
White nipple sign;
XIth nerve paralysis;X-ray hepatobiliary abnormal;
Young's syndrome;
________________________________________________________________________
Zika virus associated Guillain Barre syndrome.
________________________________________________________________________
________________________________________________________________________
An early warning prooven true;
Source:
________________________________________________________________________
In other words, carefully planned
Bio-Terrorism.
________________________________________________________________________






















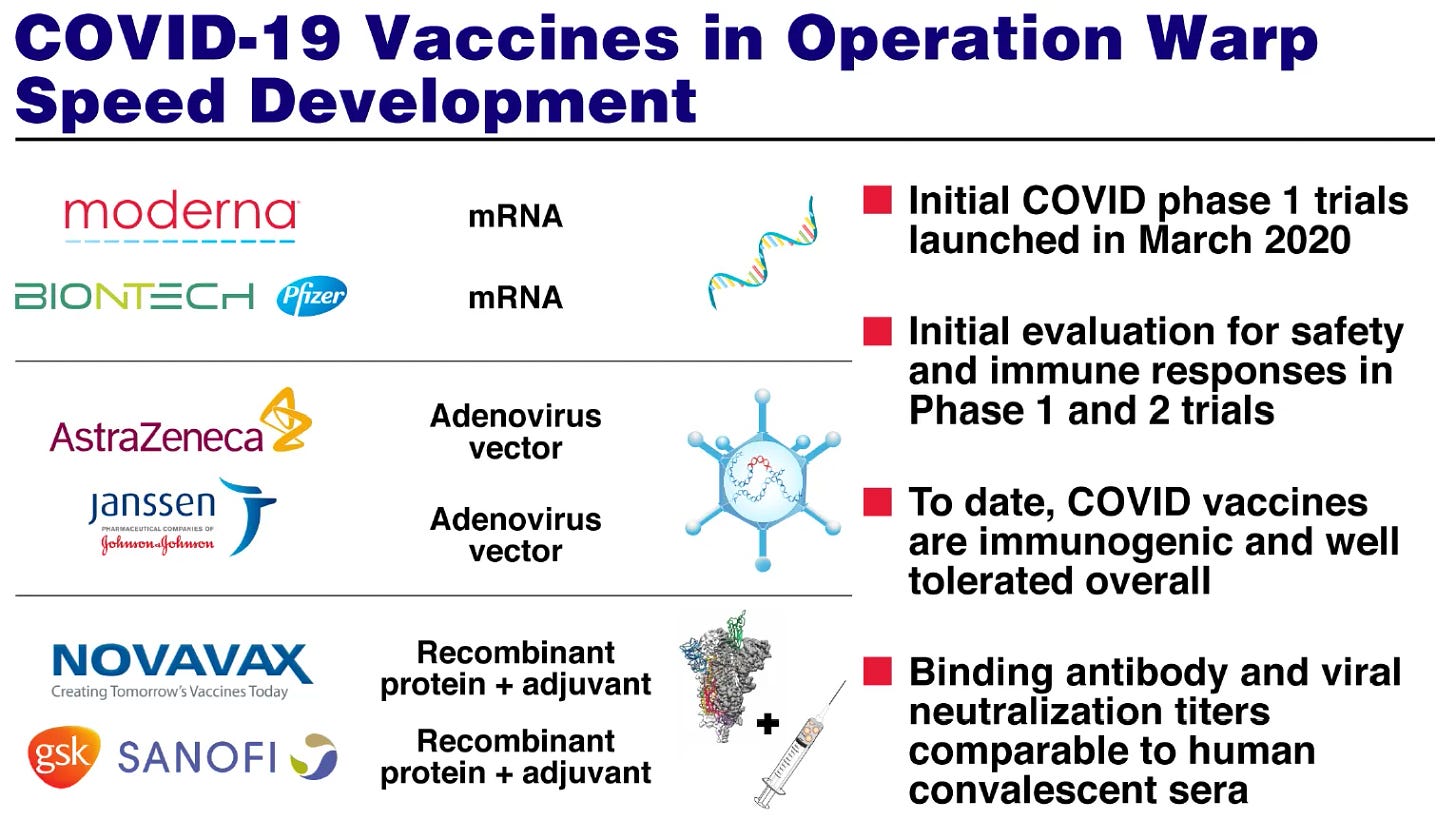











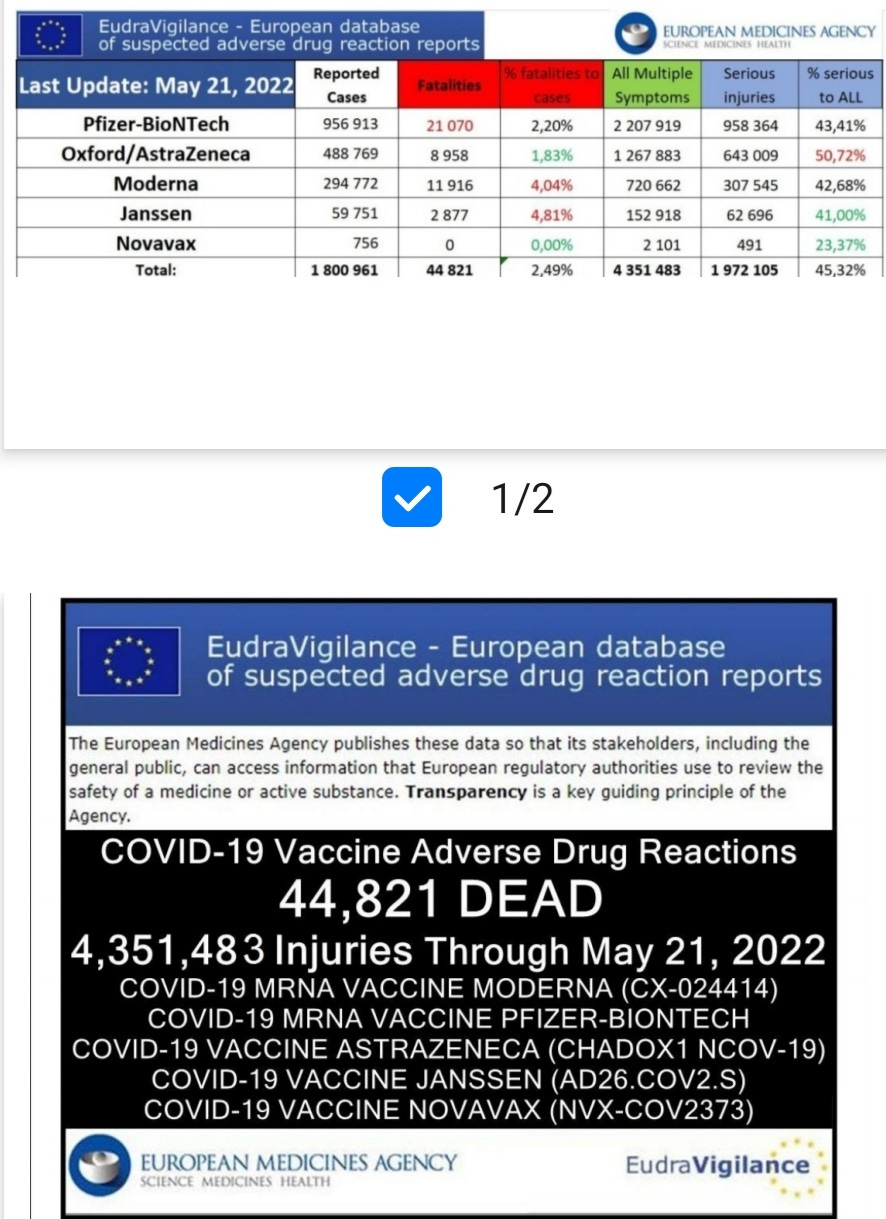








Bra arbeid Kjell Tore!
https://www.howestreet.com/2022/03/bayer-president-the-mrna-vaccines-are-gene-therapy/